From Surf Wiki (app.surf) — the open knowledge base
Tungsten borides
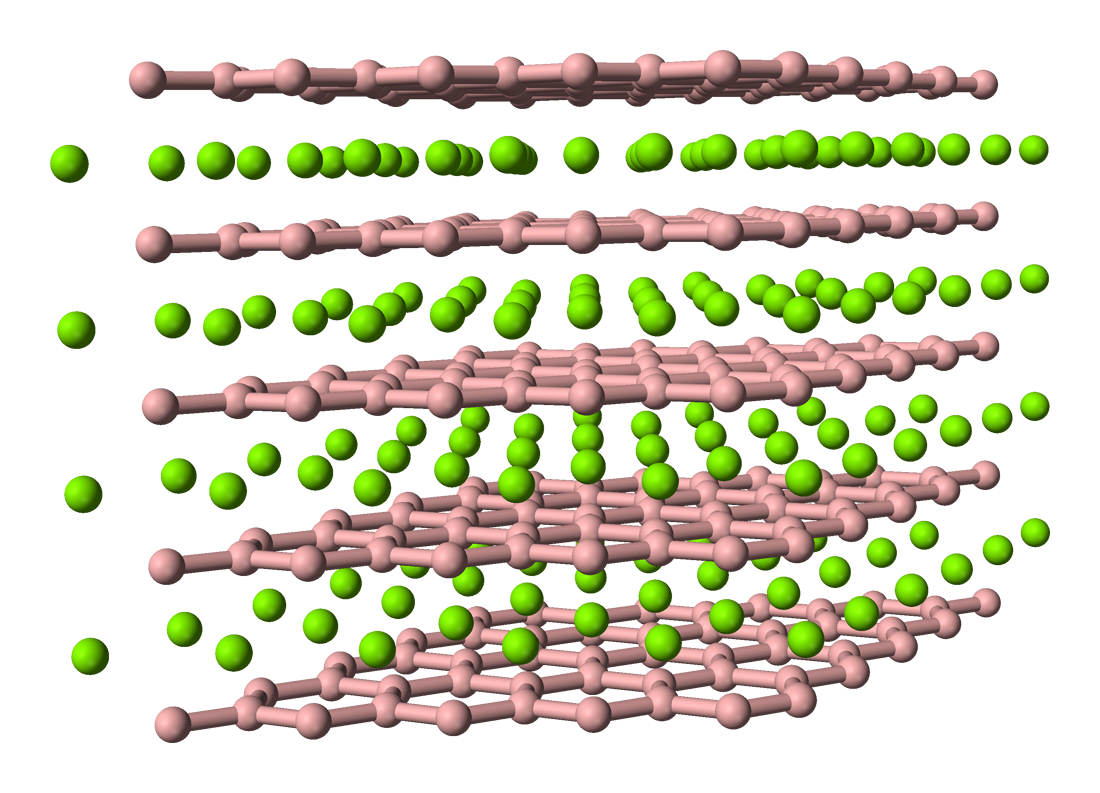

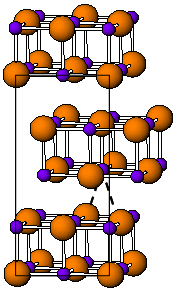
Tungsten borides are compounds of tungsten and boron. Their most remarkable property is high hardness. The Vickers hardness of WB or WB2 crystals is ~20 GPa and that of WB4 is ~30 GPa for loads exceeding 3 N.
Synthesis
Single crystals of WB2−x, x = 0.07–0.17 (about 1 cm diameter, 6 cm length) were produced by the floating zone method, and WB4 crystals can be grown by arc-melting a mixture of elemental tungsten and boron.
Structure
WB2 has the same hexagonal structure as most diborides (AlB2, MgB2, etc.). WB has several forms, α (tetragonal), β (orthorhombic) and δ (tetragonal).
Properties
δ-WB and WB2 crystals have metallic resistivities of 0.1 and 0.3 mΩ·cm, respectively. The oxidation of W2B, WB and WB2 is significant at temperatures above 600 °C. The final oxidation products contain WO3 and probably amorphous B2O3 or H3BO3. The melting temperatures of W2B, WB and WB2 are 2670, 2655 and 2365 °C, respectively.
| Material | Vickers hardness (GPa) | Bulk Modulus (GPa) | Melting point (°C) |
|---|---|---|---|
| W2B | 2670 | ||
| WB | ~20 | 2655 | |
| WB2 | ~20 | 2365 | |
| WB4 | ~30 |
References
References
- (1995). "Preparations and Some Properties of W2B, δ-WB and WB2 Crystals from High-Temperature Metal Solutions". Japanese Journal of Applied Physics.
- (1995). "Preparation of WB2−x single crystals by the floating zone method". Journal of Crystal Growth.
- (2011). "Tungsten tetraboride, an inexpensive superhard material". Proceedings of the National Academy of Sciences.
- (1966). "Tungsten Diboride: Preparation and Structure". Science.
This article was imported from Wikipedia and is available under the Creative Commons Attribution-ShareAlike 4.0 License. Content has been adapted to SurfDoc format. Original contributors can be found on the article history page.
Ask Mako anything about Tungsten borides — get instant answers, deeper analysis, and related topics.
Research with MakoFree with your Surf account
Create a free account to save articles, ask Mako questions, and organize your research.
Sign up freeThis content may have been generated or modified by AI. CloudSurf Software LLC is not responsible for the accuracy, completeness, or reliability of AI-generated content. Always verify important information from primary sources.
Report