From Surf Wiki (app.surf) — the open knowledge base
TNF inhibitor
Class of immunosuppressive drug
Class of immunosuppressive drug

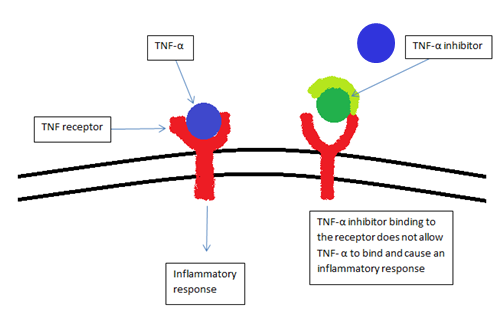
A TNF inhibitor is a pharmaceutical drug that suppresses the physiologic response to tumor necrosis factor (TNF), which is part of the inflammatory response. TNF is involved in autoimmune and immune-mediated disorders such as rheumatoid arthritis, ankylosing spondylitis, inflammatory bowel disease, psoriasis, hidradenitis suppurativa and refractory asthma, so TNF inhibitors may be used in their treatment. The important side effects of TNF inhibitors include lymphomas, infections (especially reactivation of latent tuberculosis), congestive heart failure, demyelinating disease, a lupus-like syndrome, induction of auto-antibodies, injection site reactions, and systemic side effects.
The global market for TNF inhibitors in 2008 was , in 2009 , and in 2024 .
Examples
Inhibition of TNF effects can be achieved with a monoclonal antibody such as infliximab, adalimumab, certolizumab pegol, and golimumab, or with a circulating receptor fusion protein such as etanercept.
While most clinically useful TNF inhibitors are monoclonal antibodies, some are simple molecules such as xanthine derivatives (e.g. pentoxifylline) and bupropion.
Thalidomide and its derivatives lenalidomide and pomalidomide are also active against TNF.
Several 5-HT2A agonist hallucinogens including (R)-DOI, TCB-2, LSD and LA-SS-Az have unexpectedly also been found to act as potent inhibitors of TNF, with DOI being the most active, showing TNF inhibition in the picomolar range, an order of magnitude more potent than its action as a hallucinogen. Similarly, many sigma-1 agonist psychoactive drugs including dextromethorphan, fluoxetine, and cocaine have also been found to inhibit TNF to varying degrees.
Medical uses
Rheumatoid arthritis
The role of TNF as a key player in the development of rheumatoid arthritis was originally demonstrated by Kollias and colleagues in proof of principle studies in transgenic animal models.
TNF levels have been shown to be raised in both the synovial fluid and synovium of patients with rheumatoid arthritis. This leads to local inflammation through the signalling of synovial cells to produce metalloproteinases and collagenase.
Clinical application of anti-TNF drugs in rheumatoid arthritis was demonstrated by Marc Feldmann and Ravinder N. Maini, who won the 2003 Lasker Award for their work. Anti-TNF compounds help eliminate abnormal B cell activity.
Therapy which combines certain anti-TNF agents such as etanercept with DMARDs such as methotrexate has been shown to be more effective at restoring quality of life to sufferers of rheumatoid arthritis than using either drug alone.
Skin disease
Clinical trials regarding the effectiveness of these drugs on hidradenitis suppurativa are ongoing.
The National Institute of Clinical Excellence (NICE) has issued guidelines for the treatment of severe psoriasis using the anti-TNF drugs etanercept and adalimumab as well as the anti-IL12/23 biological treatment ustekinumab. In cases where more conventional systemic treatments such as psoralen combined with ultraviolet A treatment (PUVA), methotrexate, and ciclosporin have failed or can not be tolerated, these newer biological agents may be prescribed. Infliximab may be used to treat severe plaque psoriasis if aforementioned treatments fail or can not be tolerated.
Gastrointestinal disease
In 2010 The National Institute of Clinical Excellence (NICE) in the UK issued guidelines for the treatment of severe Crohn's Disease with infliximab and adalimumab.
Cancer
Anti-TNF therapy has shown only modest effects in cancer therapy. Treatment of renal cell carcinoma with infliximab resulted in prolonged disease stabilization in certain patients. Etanercept was tested for treating patients with breast cancer and ovarian cancer showing prolonged disease stabilization in certain patients via downregulation of IL-6 and CCL2. On the other hand, adding infliximab or etanercept to gemcitabine for treating patients with advanced pancreatic cancer was not associated with differences in efficacy when compared with placebo.
Side effects
Cancer
The U.S. Food and Drug Administration continues to receive reports of a rare cancer of white blood cells (known as hepatosplenic T-cell lymphoma or HSTCL), primarily in adolescents and young adults being treated for Crohn's disease and ulcerative colitis with TNF blockers, as well as with azathioprine, and/or mercaptopurine.
Opportunistic infections
TNF inhibitors put patients at increased risk of certain opportunistic infections. The FDA has warned about the risk of infection from two bacterial pathogens, Legionella and Listeria. People taking TNF blockers are at increased risk for developing serious infections that may lead to hospitalization or death due to certain bacterial, mycobacterial, fungal, viral, and parasitic opportunistic pathogens.
Tuberculosis
In patients with latent Mycobacterium tuberculosis infection, active tuberculosis (TB) may develop soon after the initiation of treatment with infliximab. Before prescribing a TNF inhibitor, physicians should screen patients for latent tuberculosis. The anti-TNF monoclonal antibody biologics infliximab, golimumab, certolizumab and adalimumab, and the fusion protein etanercept, which are all currently approved by the FDA for human use, have warnings which state that patients should be evaluated for latent TB infection, and if it is detected, preventive treatment should be initiated prior to starting therapy with these medications.
Fungal infections
The FDA issued a warning on September 4, 2008, that patients on TNF inhibitors are at increased risk of opportunistic fungal infections such as pulmonary and disseminated histoplasmosis, coccidioidomycosis, and blastomycosis. They encourage clinicians to consider empiric antifungal therapy in certain circumstances to all patients at risk until the pathogen is identified. A recent review showed that anti-TNFα agents associate with increased infection risks for both endemic and opportunistic invasive fungal infections, particularly when given late in the overall course of treatment of the underlying disease, and in young patients receiving concomitant cytotoxic or augmented immunosuppressive therapy.
Multiple sclerosis and demyelinating disorders
In 1999 a randomized control trial was conducted testing a TNF-alpha inhibitor prototype, Lenercept, for the treatment of multiple sclerosis (MS). However, the patients in the study who received the drug had significantly more exacerbations and earlier exacerbations of their disease than those who did not.
Case reports have also come out suggesting the possibility that anti-TNF-alpha agents not only may worsen, but may cause new-onset Multiple Sclerosis or other demyelinating disorders in some patients. A 2018 case report described an Italian man with plaque psoriasis who developed MS after starting entanercept. Their literature review at that time identified 34 other cases of demyelinating disease developing after the initiation of an anti-TNF drug. Thus, anti-TNF-alpha drugs are contraindicated in patients with MS, and the American Academy of Dermatology recommends avoiding their use in those with a first degree relative with MS.
Several other monoclonal antibodies like adalimumab, pembrolizumab, nivolumab, and infliximab have been reported to trigger MS as an adverse event.
The risk of anti-TNF-associated demyelination is not associated with genetic variants of multiple sclerosis. In some studies, there were clinical differences to multiple sclerosis as 70% of the patients with anti-TNF-induced demyelination. The symptoms of demyelination do not resolve with corticosteroids, intravenous immunoglobulin or plasma exchange, and is not clear whether MS therapies are effective in anti-TNF-induced demyelination.
Paradoxical psoriasis
Despite their good safety profile, one of the reported side effects of TNF-α inhibitors is the occurrence of paradoxical psoriasis; defined as the development of psoriatic lesions or as an exacerbation of pre-existent lesions, in patients with or without a prior history of psoriasis, while undergoing treatment with TNF-α inhibitors, such as infliximab, adalimumab, and etanercept for their underlying inflammatory disease. The first case of paradoxical psoriasis induced by TNF-α inhibitors was reported in a patient with inflammatory bowel disease. Since then, an increasing number of cases have been reported in IBD cohorts and in patients suffering from other chronic immune-mediated inflammatory diseases such as rheumatoid arthritis. This increase is positively correlated with the increasing use of TNF-α inhibitors. The rates of paradoxical psoriasis reported across observational studies range from 2% to 5%, with higher rates observed in female patients. The time to onset from induction can range anywhere from a few days to a few months. The most common clinical presentations being pustular psoriasis, plaque psoriasis and guttate psoriasis, with nail and scalp involvement. Some patients may experience more than one type of lesion and/or have lesions across multiple locations.
Anti-TNF agents in nature
TNF or its effects are inhibited by several natural compounds, including curcumin (a compound present in turmeric), and catechins (in green tea). Cannabidiol and Echinacea purpurea also seem to have anti-inflammatory properties through inhibition of TNF-α production, although this effect may be mediated through cannabinoid CB1 or CB2 receptor-independent effects.
5-HT2A receptor agonists as well as sigma-1 receptor agonists have also been shown to have potent inhibitory effects on TNF-α, including psilocybin found in many species of mushrooms and DMT found in a wide variety of plants.
Thymoquinone, a compound found in the flower Nigella sativa, has been studied for possible TNF-α inhibition and related benefits for autoimmune disorder treatment.
History
Early experiments associated TNF with the pathogenesis of bacterial sepsis. Thus, the first preclinical studies using polyclonal antibodies against TNF-alpha were performed in animal models of sepsis in 1985 and showed that anti-TNF antibodies protected mice from sepsis. However, subsequent clinical trials in patients with sepsis showed no significant benefit. It wasn't until 1991 that studies in a transgenic mouse model of overexpressed human TNF provided the pre-clinical rationale for a causal role of TNF in the development of polyarthritis and that anti-TNF treatments could be effective against human arthritides. This was later confirmed in clinical trials and led to the development of the first biological therapies for rheumatoid arthritis.
References
References
- (September 2004). "A comprehensive review and evaluation of the side effects of the tumor necrosis factor alpha blockers etanercept, infliximab and adalimumab". The Journal of Dermatological Treatment.
- (September 2009). "Golimumab". Nature Reviews. Drug Discovery.
- "Top Ten/Twenty Best Selling Drugs 2009".
- "Tumor Necrosis Factor Inhibitors Market Statistics - 2035".
- (May 2002). "Binding and functional comparisons of two types of tumor necrosis factor antagonists". The Journal of Pharmacology and Experimental Therapeutics.
- (November 2001). "Cyclic nucleotide phosphodiesterases". The Journal of Allergy and Clinical Immunology.
- (February 1999). "Pentoxifylline inhibits TNF-alpha production from human alveolar macrophages". American Journal of Respiratory and Critical Care Medicine.
- (June 2006). "A new chapter opens in anti-inflammatory treatments: the antidepressant bupropion lowers production of tumor necrosis factor-alpha and interferon-gamma in mice". International Immunopharmacology.
- (December 1998). "Serotonin 5-HT2A receptor activation inhibits cytokine-stimulated inducible nitric oxide synthase in C6 glioma cells". Annals of the New York Academy of Sciences.
- (November 2008). "Serotonin 5-hydroxytryptamine(2A) receptor activation suppresses tumor necrosis factor-alpha-induced inflammation with extraordinary potency". The Journal of Pharmacology and Experimental Therapeutics.
- (December 2009). "Wishing away inflammation? New links between serotonin and TNF signaling". Molecular Interventions.
- (2013). "Dextromethorphan inhibits activations and functions in dendritic cells". Clinical & Developmental Immunology.
- (July 2020). "Inflammatory modulation of fluoxetine use in patients with depression: A systematic review and meta-analysis". Cytokine.
- (2022). "Cocaine administration protects gut mucosa barrier and reduces plasma level of TNF-α". Current Psychopharmacology.
- (December 1991). "Transgenic mice expressing human tumour necrosis factor: a predictive genetic model of arthritis". The EMBO Journal.
- (June 2015). "Regulation of tumour necrosis factor signalling: live or let die". Nature Reviews. Immunology.
- (March 2013). "TNF inhibitor therapy for rheumatoid arthritis". Biomedical Reports.
- (October 2003). "Lasker Clinical Medical Research Award. TNF defined as a therapeutic target for rheumatoid arthritis and other autoimmune diseases". Nature Medicine.
- (January 2008). "Cutting edge: anti-tumor necrosis factor therapy in rheumatoid arthritis inhibits memory B lymphocytes via effects on lymphoid germinal centers and follicular dendritic cell networks". Journal of Immunology.
- "A new view of drugs used to treat rheumatoid arthritis from medicineworld.org".
- (November 2009). "Treatment of hidradenitis suppurativa with tumour necrosis factor-alpha inhibitors". Acta Dermato-Venereologica.
- "Psoriasis Association".
- (19 May 2010). "Infliximab and adalimumab for the treatment of Crohn's disease {{!}} 1-guidance {{!}} Guidance and guidelines {{!}} NICE".
- (January 2017). "TLR-signaling and proinflammatory cytokines as drivers of tumorigenesis". Cytokine.
- "FDA Alert: Tumor Necrosis Factor (TNF) blockers, Azathioprine and/or Mercaptopurine: Update on Reports of Hepatosplenic T-Cell Lymphoma in Adolescents and Young Adults".
- "FDA Alert: Tumor Necrosis Factor-alpha (TNFα) Blockers: Label Change - Boxed Warning Updated for Risk of Infection from Legionella and Listeria".
- (October 2001). "Tuberculosis associated with infliximab, a tumor necrosis factor alpha-neutralizing agent". The New England Journal of Medicine.
- (September 4, 2008). "FDA: Manufacturers of TNF-Blocker Drugs Must Highlight Risk of Fungal Infections". [[Food and Drug Administration (United States).
- (April 2017). "Invasive fungal infections in pediatric patients treated with tumor necrosis alpha (TNF-α) inhibitors". Mycoses.
- (August 1999). "TNF neutralization in MS: results of a randomized, placebo-controlled multicenter study. The Lenercept Multiple Sclerosis Study Group and The University of British Columbia MS/MRI Analysis Group". Neurology.
- (April 2017). "CNS Demyelination with TNF-α Blockers". Current Neurology and Neuroscience Reports.
- (August 2018). "Multiple sclerosis following anti-tumor necrosis factor-alpha therapy for psoriasis: first case in Italy?". Giornale Italiano di Dermatologia e Venereologia.
- (August 2015). "Tumor Necrosis Factor-α Inhibitor Use in Psoriasis Patients With a First-degree Relative With Multiple Sclerosis". Journal of Drugs in Dermatology.
- (2 January 2020). "PPMS onset upon adalimumab treatment extends the spectrum of anti-TNF-α therapy-associated demyelinating disorders". Therapeutic Advances in Neurological Disorders.
- (2016-10-20). "Adalimumab Induced or Provoked MS in Patient with Autoimmune Uveitis: A Case Report and Review of the Literature". Case Reports in Medicine.
- (December 2019). "Multiple sclerosis associated with pembrolizumab in a patient with non-small cell lung cancer". Journal of Neurology.
- (August 2020). "Pathological findings in central nervous system demyelination associated with infliximab". Multiple Sclerosis.
- (October 2019). "Multiple sclerosis outcomes after cancer immunotherapy". Clinical & Translational Oncology.
- (December 2020). "Demyelination After Anti-TNF Therapy: Who is at Risk?". Journal of Crohn's & Colitis.
- (January 2020). "Paradoxical Psoriasis Induced by Anti-TNFα Treatment: Evaluation of Disease-Specific Clinical and Genetic Markers". International Journal of Molecular Sciences.
- (September–October 2016). "Paradoxical psoriasis after the use of anti-TNF in a patient with rheumatoid arthritis". Anais Brasileiros de Dermatologia.
- (2016-07-01). "Paradoxical reactions under TNF-α blocking agents and other biological agents given for chronic immune-mediated diseases: an analytical and comprehensive overview". RMD Open.
- (July 2006). "The anti-inflammatory effect of curcumin in an experimental model of sepsis is mediated by up-regulation of peroxisome proliferator-activated receptor-gamma". Critical Care Medicine.
- (July 2006). "Curcumin protects against radiation-induced acute and chronic cutaneous toxicity in mice and decreases mRNA expression of inflammatory and fibrogenic cytokines". International Journal of Radiation Oncology, Biology, Physics.
- (February 2006). "Effects of curcumin on tumour necrosis factor-alpha and interleukin-6 in the late phase of experimental acute pancreatitis". Journal of Veterinary Medicine. A, Physiology, Pathology, Clinical Medicine.
- (June 2005). "The effect of turmeric extracts on inflammatory mediator production". Phytomedicine.
- (August 2007). "Cannabidiol--recent advances". Chemistry & Biodiversity.
- (May 2006). "Alkylamides from Echinacea are a new class of cannabinomimetics. Cannabinoid type 2 receptor-dependent and -independent immunomodulatory effects". The Journal of Biological Chemistry.
- (2021). "Anti-Inflammatory Effects of Four Psilocybin-Containing Magic Mushroom Water Extracts in vitro on 15-Lipoxygenase Activity and on Lipopolysaccharide-Induced Cyclooxygenase-2 and Inflammatory Cytokines in Human U937 Macrophage Cells". Journal of Inflammation Research.
- (2013-10-02). "Serotonin 5-HT2A receptor activation blocks TNF-α mediated inflammation in vivo". PLOS ONE.
- (2014). "Psychedelic N,N-dimethyltryptamine and 5-methoxy-N,N-dimethyltryptamine modulate innate and adaptive inflammatory responses through the sigma-1 receptor of human monocyte-derived dendritic cells". PLOS ONE.
- (September 2015). "Thymoquinone inhibits TNF-α-induced inflammation and cell adhesion in rheumatoid arthritis synovial fibroblasts by ASK1 regulation". Toxicology and Applied Pharmacology.
- (2018). "The Neuroprotective Effects of Thymoquinone: A Review". Dose-Response.
- (February 2021). "Thymoquinone in autoimmune diseases: Therapeutic potential and molecular mechanisms". Biomedicine & Pharmacotherapy.
- (November 2021). "Thymoquinone: A small molecule from nature with high therapeutic potential". Drug Discovery Today.
- (July 2008). "First demonstration of the role of TNF in the pathogenesis of disease". Journal of Immunology.
- (July 2008). "Passive immunization against cachectin/tumor necrosis factor protects mice from lethal effect of endotoxin. Science, 1985, 229(4716):869-871. Classical article". Journal of Immunology.
- (October 1994). "Randomised double-blind comparison of chimeric monoclonal antibody to tumour necrosis factor alpha (cA2) versus placebo in rheumatoid arthritis". Lancet.
This article was imported from Wikipedia and is available under the Creative Commons Attribution-ShareAlike 4.0 License. Content has been adapted to SurfDoc format. Original contributors can be found on the article history page.
Ask Mako anything about TNF inhibitor — get instant answers, deeper analysis, and related topics.
Research with MakoFree with your Surf account
Create a free account to save articles, ask Mako questions, and organize your research.
Sign up freeThis content may have been generated or modified by AI. CloudSurf Software LLC is not responsible for the accuracy, completeness, or reliability of AI-generated content. Always verify important information from primary sources.
Report