From Surf Wiki (app.surf) — the open knowledge base
Thyroglobulin
Protein produced and used by the thyroid
Protein produced and used by the thyroid
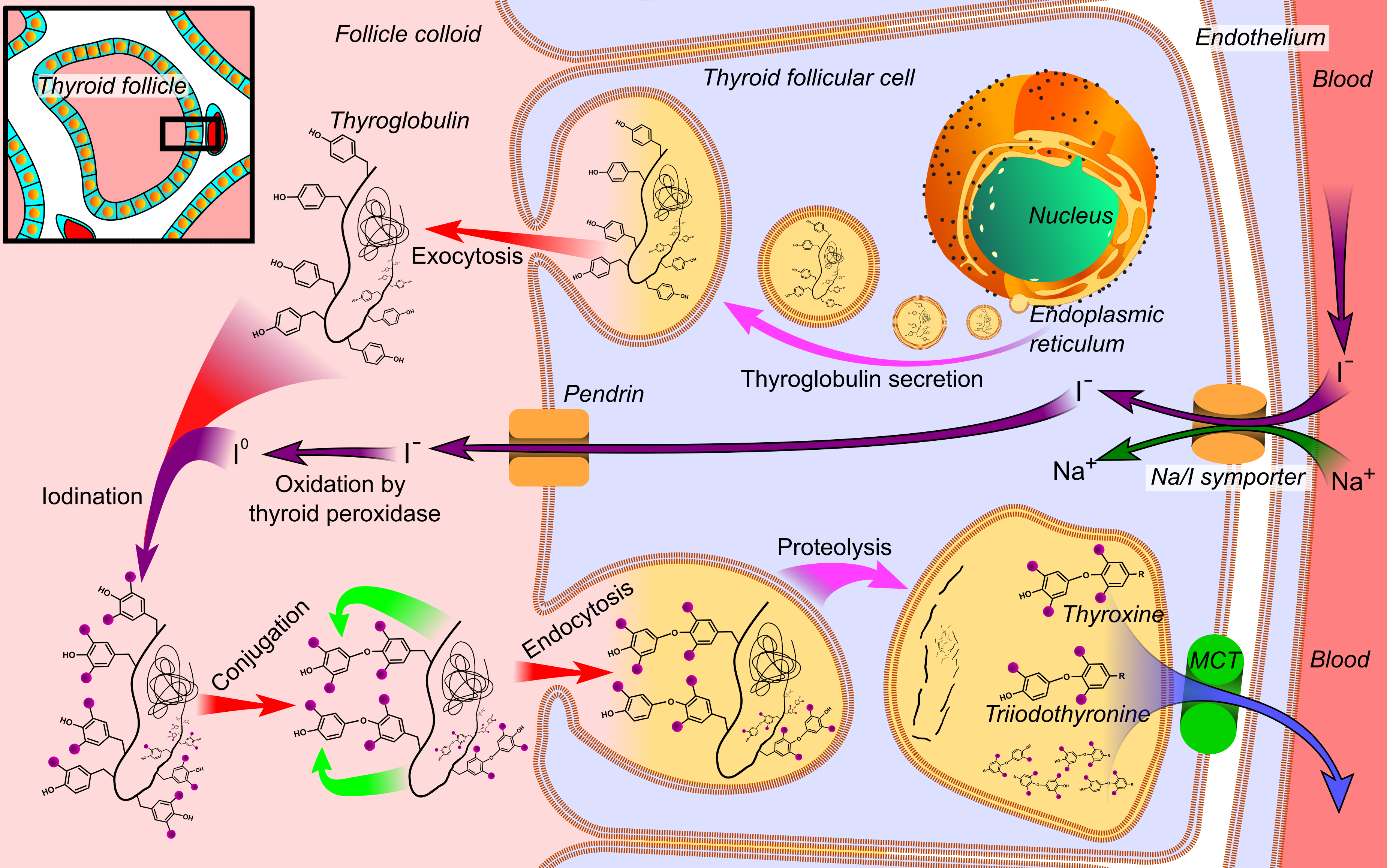
Thyroglobulin (Tg) is a 660 kDa, dimeric glycoprotein produced by the follicular cells of the thyroid and used entirely within the thyroid gland. Tg is secreted and accumulated at hundreds of grams per litre in the extracellular compartment of the thyroid follicles, accounting for approximately half of the protein content of the thyroid gland. Human TG (hTG) is a homodimer of subunits each containing 2768 amino acids as synthesized (a short signal peptide of 19 amino acids may be removed from the N-terminus in the mature protein).
Thyroglobulin is in all vertebrates the main precursor to thyroid hormones, which are produced when thyroglobulin's tyrosine residues are combined with iodine and the protein is subsequently cleaved. Each thyroglobulin molecule contains approximately 16 tyrosine residues, but only around 10 of these are subject to iodination by thyroperoxidase in the follicular colloid. It takes two iodinated tyrosines to make a thyroid hormone molecule; therefore, each Tg molecule forms approximately 5 thyroid hormone molecules.
Function

Thyroglobulin (Tg) acts as a substrate for the synthesis of the thyroid hormones thyroxine (T4) and triiodothyronine (T3), as well as the storage of the inactive forms of thyroid hormone and iodine within the follicular lumen of a thyroid follicle.
Newly synthesized thyroid hormones (T3 and T4) exist as prohormonal modified tyrosine residues of thyroglobulin, which comprise the colloid within the follicle. When stimulated by thyroid stimulating hormone (TSH), the colloid of the follicular lumen is endocytosed into the surrounding thyroid follicular epithelial cells. When endosomes fuse with lysosomes in these cells, Tg is subsequently cleaved by proteases to release thyroglobulin from its T3 and T4 attachments.
Because of the hydrophobic nature of T3 and T4, they are able to then diffuse across the lysosomal membrane and into the cytoplasm. The active forms of thyroid hormone (T3 and T4), are then released into circulation where they are either unbound or attached to plasma proteins. Iodine is recycled back into the follicular lumen where it can continue to serve as a substrate for thyroid hormone synthesis, while the non-modified residues of Tg reenter cellular metabolic pathways.
Clinical significance
Half-life and clinical elevation
Metabolism of thyroglobulin occurs in the liver via thyroid gland recycling of the protein. Circulating thyroglobulin has a half-life of 65 hours. Following thyroidectomy, it may take many weeks before thyroglobulin levels become undetectable. Thyroglobulin levels may be tested regularly for a few weeks or months following the removal of the thyroid. After thyroglobulin levels become undetectable (following thyroidectomy), levels can be serially monitored in follow-up of patients with papillary or follicular thyroid carcinoma.
A subsequent elevation of the thyroglobulin level is an indication of recurrence of papillary or follicular thyroid carcinoma. In other words, a rise in thyroglobulin levels in the blood may be a sign that thyroid cancer cells are growing and/or the cancer is spreading. Hence, thyroglobulin levels in the blood are mainly used as a tumor marker for certain kinds of thyroid cancer (particularly papillary or follicular thyroid cancer). Thyroglobulin is not produced by medullary or anaplastic thyroid carcinoma.
Thyroglobulin levels are tested via blood tests including immunoassays and highly-sensitive thyroglobulin (hsTg) assays. These tests are often ordered after thyroid cancer treatment.
Thyroglobulin antibodies
In the clinical laboratory, thyroglobulin testing can be complicated by the presence of anti-thyroglobulin antibodies (ATAs, alternatively referred to as TgAb). Anti-thyroglobulin antibodies are present in 1 in 10 normal individuals, and a greater percentage of patients with thyroid carcinoma. The presence of these antibodies can result in falsely low (or rarely falsely high) levels of reported thyroglobulin, a problem that can be somewhat circumvented by concomitant testing for the presence of ATAs. The ideal strategy for a clinician's interpretation and management of patient care in the event of confounding detection of ATAs is testing to follow serial quantitative measurements (rather than a single laboratory measurement).
ATAs are often found in patients with Hashimoto's thyroiditis or Graves' disease. Their presence is of limited use in the diagnosis of these diseases, since they may also be present in healthy euthyroid individuals. ATAs are also found in patients with Hashimoto's encephalopathy, a neuroendocrine disorder related to—but not caused by—Hashimoto's thyroiditis.
Interactions
Thyroglobulin has been shown to interact with Binding immunoglobulin protein.
References
References
- Boron WF. (2003). "Medical Physiology: A Cellular And Molecular Approach". Elsevier/Saunders.
- "Protein" thyroglobulin precursor [Homo sapiens]". National Center for Biotechnology Information, U.S. National Library of Medicine.
- "TG thyroglobulin [Homo sapiens (human)] – Gene – NCBI".
- (June 2022). "A glance at post-translational modifications of human thyroglobulin: potential impact on function and pathogenesis". European Thyroid Journal.
- (2021-01-01). "The effect of specific binding proteins on immunoassay measurements of total and free thyroid hormones and cortisol". Therapeutic Advances in Endocrinology and Metabolism.
- (2000). "Endotext". MDText.com, Inc..
- "Thyroglobulin: MedlinePlus Lab Test Information".
- (November 2021). "Life after thyroid cancer: the role of thyroglobulin and thyroglobulin antibodies for postoperative follow-up". Expert Review of Endocrinology & Metabolism.
- "ACS :: Tumor Markers". American Cancer Society.
- (December 2019). "Unstimulated high-sensitive thyroglobulin is a powerful prognostic predictor in patients with thyroid cancer". Clinical Chemistry and Laboratory Medicine.
- (February 2003). "Antithyroid antibodies in the CSF: their role in the pathogenesis of Hashimoto's encephalopathy". Neurology.
- (June 2001). "Role of extracellular molecular chaperones in the folding of oxidized proteins. Refolding of colloidal thyroglobulin by protein disulfide isomerase and immunoglobulin heavy chain-binding protein". The Journal of Biological Chemistry.
- (February 1999). "Involvement of oxidative reactions and extracellular protein chaperones in the rescue of misassembled thyroglobulin in the follicular lumen". Biochemical and Biophysical Research Communications.
This article was imported from Wikipedia and is available under the Creative Commons Attribution-ShareAlike 4.0 License. Content has been adapted to SurfDoc format. Original contributors can be found on the article history page.
Ask Mako anything about Thyroglobulin — get instant answers, deeper analysis, and related topics.
Research with MakoFree with your Surf account
Create a free account to save articles, ask Mako questions, and organize your research.
Sign up freeThis content may have been generated or modified by AI. CloudSurf Software LLC is not responsible for the accuracy, completeness, or reliability of AI-generated content. Always verify important information from primary sources.
Report