From Surf Wiki (app.surf) — the open knowledge base
Thorpe reaction
Chemical reaction
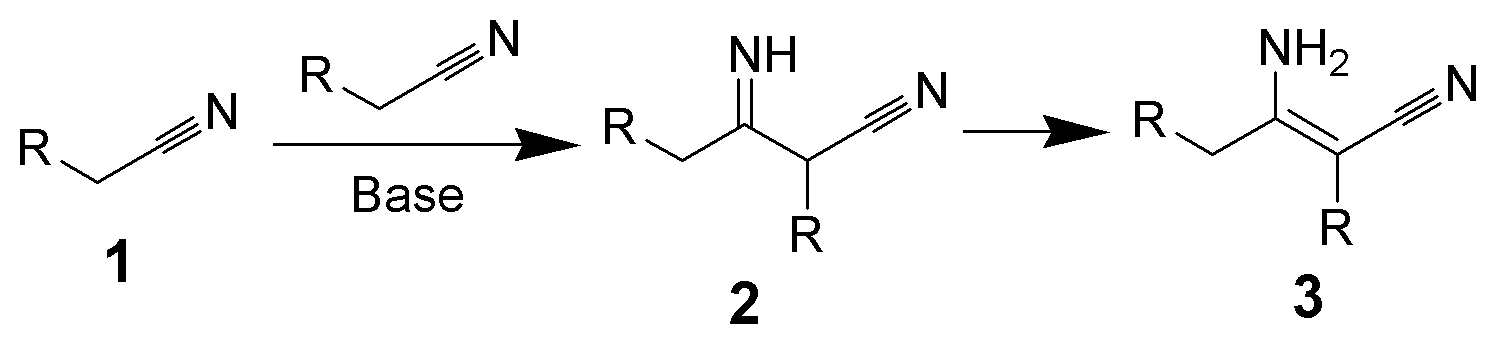
Chemical reaction
The Thorpe reaction is a chemical reaction described as a self-condensation of aliphatic nitriles catalyzed by base to form enamines. The reaction was discovered by Jocelyn Field Thorpe.

Thorpe–Ziegler reaction
The Thorpe–Ziegler reaction (named after Jocelyn Field Thorpe and Karl Ziegler), or Ziegler method, is the intramolecular modification with a dinitrile as a reactant and a cyclic ketone as the final reaction product after acidic hydrolysis. The reaction is conceptually related to the Dieckmann condensation.
References
References
- (1904). "CLXXV.—The formation and reactions of imino-compounds. Part I. Condensation of ethyl cyanoacetate with its sodium derivative". Journal of the Chemical Society, Transactions.
- (1933). "Über vielgliedrige Ringsysteme. I. Die präparativ ergiebige Synthese der Polymethylenketone mit mehr als 6 Ringgliedern". Justus Liebig's Annalen der Chemie.
- (2011). "Organic Reactions".
This article was imported from Wikipedia and is available under the Creative Commons Attribution-ShareAlike 4.0 License. Content has been adapted to SurfDoc format. Original contributors can be found on the article history page.
Ask Mako anything about Thorpe reaction — get instant answers, deeper analysis, and related topics.
Research with MakoFree with your Surf account
Create a free account to save articles, ask Mako questions, and organize your research.
Sign up freeThis content may have been generated or modified by AI. CloudSurf Software LLC is not responsible for the accuracy, completeness, or reliability of AI-generated content. Always verify important information from primary sources.
Report