From Surf Wiki (app.surf) — the open knowledge base
Sharpless epoxidation
Chemical reaction
Chemical reaction
The Sharpless epoxidation reaction is an enantioselective chemical reaction to prepare 2,3-epoxyalcohols from primary and secondary allylic alcohols. The oxidizing agent is tert-butyl hydroperoxide. The method relies on a catalyst formed from titanium tetra(isopropoxide) and diethyl tartrate.
2,3-Epoxyalcohols can be converted into diols, aminoalcohols, and ethers. The reactants for the Sharpless epoxidation are commercially available and relatively inexpensive. K. Barry Sharpless published a paper on the reaction in 1980 and was awarded the 2001 Nobel Prize in Chemistry for this and related work on asymmetric oxidations. The prize was shared with William S. Knowles and Ryōji Noyori.
Catalyst
5–10 mol% of the catalyst is typical. The presence of 3Å molecular sieves (3Å MS) is necessary.*{{cite journal|author1=Hill, J. G. |author2=Sharpless, K. B. |author3=Exon, C. M. |author4=Regenye, R. |journal=Org. Synth.|volume=63|pages=66|year=1985|doi=10.15227/orgsyn.063.0066| title=Enantioselective Epoxidation of Allylic Alcohols: (2s,3s)-3-propyloxiranemethanol}} The structure of the catalyst is uncertain although it is thought to be a dimer of [Ti(tartrate)(OR)].
Selectivity
The epoxidation of allylic alcohols is a well-utilized conversion in fine chemical synthesis. The chirality of the product of a Sharpless epoxidation is sometimes predicted with the following mnemonic. A rectangle is drawn around the double bond in the same plane as the carbons of the double bond (the xy-plane), with the allylic alcohol in the bottom right corner and the other substituents in their appropriate corners. In this orientation, the (−) diester tartrate preferentially interacts with the top half of the molecule, and the (+) diester tartrate preferentially interacts with the bottom half of the molecule. This model seems to be valid despite substitution on the olefin. Selectivity decreases with larger R1, but increases with larger R2 and R3 (see introduction).
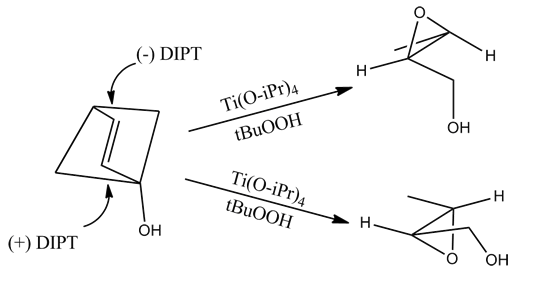
However, this method incorrectly predicts the product of allylic 1,2-diols.

Kinetic resolution
The Sharpless epoxidation can also give kinetic resolution of a racemic mixture of secondary 2,3-epoxyalcohols. While the yield of a kinetic resolution process cannot be higher than 50%, the enantiomeric excess approaches 100% in some reactions.

Synthetic utility
The Sharpless epoxidation is viable with a large range of primary and secondary alkenic alcohols. Furthermore, with the exception noted above, a given dialkyl tartrate will preferentially add to the same face independent of the substitution on the alkene.To demonstrate the synthetic utility of the Sharpless epoxidation, the Sharpless group created synthetic intermediates of various natural products: methymycin, erythromycin, leukotriene C-1, and (+)-disparlure.

As one of the few highly enantioselective reactions during its time, many manipulations of the 2,3-epoxyalcohols have been developed.
The Sharpless epoxidation has been used for the total synthesis of various saccharides, terpenes, leukotrienes, pheromones, and antibiotics.
The main drawback of this protocol is the necessity of the presence of an allylic alcohol. The Jacobsen epoxidation, an alternative method to enantioselectively oxidise alkenes, overcomes this issue and tolerates a wider array of functional groups. For specifically glycidic epoxides, the Jørgensen-Córdova epoxidation avoids the need to reduce the carbonyl and then reoxidize, and has more efficient catalyst turnover.
References of historic interest
References
References
- Diego J. Ramón and Miguel Yus. (2006). "In the Arena of Enantioselective Synthesis, Titanium Complexes Wear the Laurel Wreath". Chem. Rev..
- Johnson, R. A.. (1991). "Addition Reactions with Formation of Carbon–Oxygen Bonds: (ii) Asymmetric Methods of Epoxidation". Compr. Org. Synth..
- Hüft, E.. (1993). "Enantioselective epoxidation with peroxidic oxygen". Top. Curr. Chem..
- (1996). "Asymmetric Epoxidation of Allylic Alcohols: The Katsuki-Sharpless Epoxidation Reaction". Org. React..
- Pfenninger, A.. (1986). "Asymmetric Epoxidation of Allylic Alcohols: The Sharpless Epoxidation". Synthesis.
- A. Pfenninger. (1986). "Asymmetric Epoxidation of Allylic Alcohols: The Sharpless Epoxidation". Synthesis.
- Finn, M. G.. (1991). "Mechanism of Asymmetric Epoxidation. 2. Catalyst Structure". J. Am. Chem. Soc..
- (1991). "Inversion of enantioselectivity in the kinetic resolution mode of the Katsuki-Sharpless asymmetric epoxidation reaction". [[J. Am. Chem. Soc.]].
- (1988). "A highly efficient kinetic resolution of γ- and β- trimethylsilyl secondary allylic alcohols by the sharpless asymmetric epoxidation". [[Tetrahedron (journal).
- Martin, V.. (1981). "Kinetic resolution of racemic allylic alcohols by enantioselective epoxidation. A route to substances of absolute enantiomeric purity?". [[J. Am. Chem. Soc.]].
- Rossiter, B.. (1981). "Asymmetric epoxidation provides shortest routes to four chiral epoxy alcohols which are key intermediates in syntheses of methymycin, erythromycin, leukotriene C-1, and disparlure". [[J. Am. Chem. Soc.]].
- [[K. Barry Sharpless. (1983). "Stereo and regioselective openings of chiral 2,3-epoxy alcohols. Versatile routes to optically pure natural products and drugs. Unusual kinetic resolutions". [[Pure Appl. Chem.]].
- Taber, Douglass F.. (5 July 2010). "The Nicolaou synthesis of (+)-Hirsutellone B". Organic Chemistry Highlights.
This article was imported from Wikipedia and is available under the Creative Commons Attribution-ShareAlike 4.0 License. Content has been adapted to SurfDoc format. Original contributors can be found on the article history page.
Ask Mako anything about Sharpless epoxidation — get instant answers, deeper analysis, and related topics.
Research with MakoFree with your Surf account
Create a free account to save articles, ask Mako questions, and organize your research.
Sign up freeThis content may have been generated or modified by AI. CloudSurf Software LLC is not responsible for the accuracy, completeness, or reliability of AI-generated content. Always verify important information from primary sources.
Report