From Surf Wiki (app.surf) — the open knowledge base
Plasmodium
Genus of parasitic protists that can cause malaria

Genus of parasitic protists that can cause malaria
- Plasmodium (Bennettinia)
- Plasmodium (Carinamoeba)
- Plasmodium carmelinoi
- Plasmodium (Giovannolaia)
- Plasmodium circumflexum
- Plasmodium homocircumflexum
- Plasmodium lophurae
- Plasmodium polare
- Plasmodium (Haemamoeba)
- Plasmodium cathemerium
- Plasmodium gallinaceum
- Plasmodium lutzi
- Plasmodium matutinum
- Plasmodium relictum
- Plasmodium tejerai
- Plasmodium (Huffia)
- Plasmodium elongatum
- Plasmodium (Lacertamoeba)
- Plasmodium floridense
- Plasmodium (Laverania)
- Plasmodium billbrayi
- Plasmodium falciparum
- Plasmodium gaboni
- Plasmodium lomamiensis
- Plasmodium reichenowi
- Plasmodium (Novyella)
- Plasmodium ashfordi
- Plasmodium delichoni
- Plasmodium globularis
- Plasmodium homonucleophilum
- Plasmodium homopolare
- Plasmodium juxtanucleare
- Plasmodium lucens
- Plasmodium megaglobularis
- Plasmodium multivacuolaris
- Plasmodium nucleophilum
- Plasmodium parahexamerium
- Plasmodium paranucleophilum
- Plasmodium rouxi
- Plasmodium unalis
- Plasmodium vaughani
- Plasmodium (Paraplasmodium)
- Plasmodium chiricahuae
- Plasmodium mexicanum
- Plasmodium (Plasmodium)
- Plasmodium brasilianum
- Plasmodium cynomolgi
- Plasmodium cynomolgi bastianelli
- Plasmodium cynomolgi ceylonensis
- Plasmodium fieldi
- Plasmodium fragile
- Plasmodium gonderi
- Plasmodium inui
- Plasmodium inui inui
- Plasmodium knowlesi
- Plasmodium malariae
- Plasmodium ovale
- Plasmodium ovale curtisi
- Plasmodium ovale wallikeri
- Plasmodium simiovale
- Plasmodium vivax
- Plasmodium (Sauramoeba)
- Plasmodium agamae
- Plasmodium giganteum
- Plasmodium kentropyxi
- Plasmodium (Vinckeia)
- Plasmodium atheruri
- Plasmodium berghei
- Plasmodium berghei ANKA
- Plasmodium berghei yoelii
- Plasmodium chabaudi
- Plasmodium chabaudi adami
- Plasmodium chabaudi chabaudi
- Plasmodium vinckei
- Plasmodium vinckei brucechwatti
- Plasmodium vinckei lentum
- Plasmodium vinckei petteri
- Plasmodium vinckei vinckei
- Plasmodium yoelii
- Plasmodium yoelii killicki
- Plasmodium yoelii nigeriensis
- Plasmodium yoelii YM
- Plasmodium yoelii yoelii
- Species incertae sedis
- Plasmodium azurophilum
- Plasmodium bubalis
- Plasmodium coatneyi
- Plasmodium cuculus
- Plasmodium cyclopsi
- Plasmodium fairchildi
- Plasmodium gemini
- Plasmodium guanggong
- Plasmodium heteronucleare
- Plasmodium hispaniolae
- Plasmodium hylobati
- Plasmodium intabazwe
- Plasmodium koreafense
- Plasmodium lacertiliae
- Plasmodium leucocytica
- Plasmodium mackerrasae
- Plasmodium megalotrypa
- Plasmodium minuoviride
- Plasmodium odocoilei
- Plasmodium simium
- Plasmodium streptopelia
- Plasmodium zonuriae --
Plasmodium is a genus of unicellular eukaryotes that are obligate parasites of vertebrates and insects. The life cycles of Plasmodium species involve development in a blood-feeding insect host which then injects parasites into a vertebrate host during a blood meal. Parasites grow within a vertebrate body tissue (often the liver) before entering the bloodstream to infect red blood cells. The ensuing destruction of host red blood cells can result in malaria. During this infection, some parasites are picked up by a blood-feeding insect (mosquitoes in majority cases), continuing the life cycle.
Plasmodium is a member of the phylum Apicomplexa, a large group of parasitic eukaryotes. Within Apicomplexa, Plasmodium is in the order Haemosporida and family Plasmodiidae. Over 200 species of Plasmodium have been described, many of which have been subdivided into 14 subgenera based on parasite morphology and host range. Evolutionary relationships among different Plasmodium species do not always follow taxonomic boundaries; some species that are morphologically similar or infect the same host turn out to be distantly related.
Species of Plasmodium are distributed globally wherever suitable hosts are found. Insect hosts are most frequently mosquitoes of the genera Culex and Anopheles. Vertebrate hosts include reptiles, birds, and mammals. Plasmodium parasites were first identified in the late 19th century by Charles Laveran. Over the course of the 20th century, many other species were discovered in various hosts and classified, including five species that regularly infect humans: P. vivax, P. falciparum, P. malariae, P. ovale, and P. knowlesi. P. falciparum is by far the most lethal in humans, resulting in hundreds of thousands of deaths per year. A number of drugs have been developed to treat Plasmodium infection; however, the parasites have evolved resistance to each drug developed.
Although the parasite can also infect people via blood transfusion, this is very rare, and Plasmodium cannot be spread from person to person. Some of subspecies of Plasmodium are obligate intracellular parasites.
Description

The genus Plasmodium consists of all eukaryotes in the phylum Apicomplexa that both undergo the asexual replication process of merogony inside host red blood cells and produce the crystalline pigment hemozoin as a byproduct of digesting host hemoglobin. Attached to the nucleus is the endoplasmic reticulum (ER), which functions similarly to the ER in other eukaryotes. Proteins are trafficked from the ER to the Golgi apparatus which generally consists of a single membrane-bound compartment in Apicomplexans. From here, proteins are trafficked to various cellular compartments or to the cell surface.
Like other apicomplexans, Plasmodium species have several cellular structures at the apical end of the parasite that serve as specialized organelles for secreting effectors into the host. The most prominent are the bulbous rhoptries which contain parasite proteins involved in invading the host cell and modifying the host once inside. Adjacent to the rhoptries are smaller structures termed micronemes that contain parasite proteins required for motility as well as recognizing and attaching to host cells. Spread throughout the parasite are secretory vesicles called dense granules that contain parasite proteins involved in modifying the membrane that separates the parasite from the host, termed the parasitophorous vacuole.
Species of Plasmodium also contain two large membrane-bound organelles of endosymbiotic origin, the mitochondrion and the apicoplast, both of which play key roles in the parasite's metabolism. Unlike mammalian cells which contain many mitochondria, Plasmodium cells contain a single large mitochondrion that coordinates its division with that of the Plasmodium cell. Like in other eukaryotes, the Plasmodium mitochondrion is capable of generating energy in the form of ATP via the citric acid cycle; however, this function is only required for parasite survival in the insect host, and is not needed for growth in red blood cells. The apicoplast is involved in the synthesis of various metabolic precursors, including fatty acids, isoprenoids, iron-sulphur clusters, and components of the heme biosynthesis pathway.
Life cycle


The life cycle of Plasmodium involves several distinct stages in the insect and vertebrate hosts. Parasites are generally introduced into a vertebrate host by the bite of an insect host (generally a mosquito, with the exception of some Plasmodium species of reptiles). Parasites first infect the liver or other tissue, where they undergo a single large round of replication before exiting the host cell to infect erythrocytes. At this point, some species of Plasmodium of primates can form a long-lived dormant stage called a hypnozoite, which can remain in the liver for more than a year. However, for most Plasmodium species, the parasites in infected liver cells are only what are called merozoites. After emerging from the liver, they enter red blood cells, as explained above. They then go through continuous cycles of erythrocyte infection, while a small percentage of parasites differentiate into a sexual stage called a gametocyte which is picked up by an insect host taking a blood meal. In some hosts, invasion of erythrocytes by Plasmodium species can result in disease, called malaria. This can sometimes be severe, rapidly followed by death of the host (e.g. P. falciparum in humans). In other hosts, Plasmodium infection can apparently be asymptomatic.
Even when humans have such subclinical plasmodial infections, there can nevertheless be very large numbers of multiplying parasites concealed in, particularly, the spleen and bone marrow. Certainly, this applies in the case of P. vivax. These hidden parasites (in addition to hypnozoites) are thought to be the origin of instances of recurrent P. vivax malaria.
.jpg)
Within the red blood cells, the merozoites grow first to a ring-shaped form and then to a larger form called a trophozoite. Trophozoites then mature to schizonts which divide several times to produce new merozoites. The infected red blood cell eventually bursts, allowing the new merozoites to travel within the bloodstream to infect new red blood cells. Most merozoites continue this replicative cycle, however some merozoites upon infecting red blood cells differentiate into male or female sexual forms called gametocytes. These gametocytes circulate in the blood until they are taken up when a mosquito feeds on the infected vertebrate host, taking up blood which includes the gametocytes.
In the mosquito, the gametocytes move along with the blood meal to the mosquito's midgut. Here the gametocytes develop into male and female gametes which fertilize each other, forming a zygote. Zygotes then develop into a motile form called an ookinete, which penetrates the wall of the midgut. Upon traversing the midgut wall, the ookinete embeds into the gut's exterior membrane and develops into an oocyst. Oocysts divide many times to produce large numbers of small elongated sporozoites. These sporozoites migrate to the salivary glands of the mosquito where they can be injected into the blood of the next host the mosquito bites, repeating the cycle.
Evolution and taxonomy

Taxonomy
Plasmodium belongs to the phylum Apicomplexa, a taxonomic group of single-celled parasites with characteristic secretory organelles at one end of the cell. Within Apicomplexa, Plasmodium is within the order Haemosporida, a group that includes all apicomplexans that live within blood cells. Based on the presence of the pigment hemozoin and the method of asexual reproduction, the order is further split into four families, of which Plasmodium is in the family Plasmodiidae.
The genus Plasmodium consists of over 200 species, generally described on the basis of their appearance in blood smears of infected vertebrates. These species have been categorized on the basis of their morphology and host range into 14 subgenera:
- Subgenus Asiamoeba (Telford, 1988) – reptiles
- Subgenus Bennettinia (Valkiunas, 1997) – birds
- Subgenus Carinamoeba (Garnham, 1966) – reptiles
- Subgenus Giovannolaia (Corradetti, et al. 1963) – birds
- Subgenus Haemamoeba (Corradetti, et al. 1963) – birds
- Subgenus Huffia (Corradetti, et al. 1963) – birds
- Subgenus Lacertamoeba (Telford, 1988) – reptiles
- Subgenus Laverania (Bray, 1958) – great apes, humans
- Subgenus Novyella (Corradetti, et al. 1963) – birds
- Subgenus Ophidiella (Telford, 1988) – reptiles
- Subgenus Paraplasmodium (Telford, 1988) – reptiles
- Subgenus Plasmodium (Bray, 1955) – monkeys and apes
- Subgenus Sauramoeba (Garnham, 1966) – reptiles
- Subgenus Vinckeia (Garnham, 1964) – mammals inc. primates
Species infecting monkeys and apes with the exceptions of P. falciparum and P. reichenowi (which together make up the subgenus Laverania) are classified in the subgenus Plasmodium. Parasites infecting other mammals including some primates (lemurs and others) are classified in the subgenus Vinckeia. The five subgenera Bennettinia, Giovannolaia, Haemamoeba, Huffia, and Novyella contain the known avian malarial species. The remaining subgenera: Asiamoeba, Carinamoeba, Lacertamoeba, Ophidiella, Paraplasmodium, and Sauramoeba contain the diverse groups of parasites found to infect reptiles.
Phylogeny
More recent studies of Plasmodium species using molecular methods have implied that the group's evolution has not perfectly followed taxonomy. Many Plasmodium species that are morphologically similar or infect the same hosts turn out to be only distantly related. In the 1990s, several studies sought to evaluate evolutionary relationships of Plasmodium species by comparing ribosomal RNA and a surface protein gene from various species, finding the human parasite P. falciparum to be more closely related to avian parasites than to other parasites of primates.
Estimates for when different Plasmodium lineages diverged have differed broadly. Estimates for the diversification of the order Haemosporida range from around 16.2 million to 100 million years ago. There has been particular interest in dating the divergence of the human parasite P. falciparum from other Plasmodium lineages due to its medical importance. For this, estimated dates range from 110,000 to 2.5 million years ago.
Distribution
Plasmodium species are distributed globally. All Plasmodium species are parasitic and must pass between a vertebrate host and an insect host to complete their life cycles. Different species of Plasmodium display different host ranges, with some species restricted to a single vertebrate and insect host, while other species can infect several species of vertebrates and/or insects.
Vertebrates
_species_by_country_of_origin_for_imported_cases_to_non-endemic_countries.png)
Plasmodium parasites have been described in a broad array of vertebrate hosts including reptiles, birds, and mammals.
Humans are primarily infected by five species of Plasmodium, with the overwhelming majority of severe disease and death caused by Plasmodium falciparum. Some species that infect humans can also infect other primates, and zoonoses of certain species (e.g. P. knowlesi) from other primates to humans are common. Many other mammals also carry Plasmodium species, such as a variety of rodents, ungulates, and bats. Again, some species of Plasmodium can cause severe disease in some of these hosts, while many appear not to.
Over 150 species of Plasmodium infect a broad variety of birds. In general each species of Plasmodium infects one to a few species of birds. Plasmodium parasites that infect birds tend to persist in a given host for years or for the life time of the host, although in some cases Plasmodium infections can result in severe illness and rapid death. Unlike with Plasmodium species infecting mammals, those infecting birds are distributed across the globe.
Species from several subgenera of Plasmodium infect diverse reptiles. Plasmodium parasites have been described in most lizard families and, like avian parasites, are spread worldwide. Again, parasites can result either in severe disease or be apparently asymptomatic depending on the parasite and the host.
A number of drugs have been developed over the years to control Plasmodium infection in vertebrate hosts, particularly in humans. Quinine was used as a frontline antimalarial from the 17th century until widespread resistance emerged in the early 20th century. Resistance to quinine spurred the development of a broad array of antimalarial medications through the 20th century including chloroquine, proguanil, atovaquone, sulfadoxine/pyrimethamine, mefloquine, and artemisinin. In general, antimalarial drugs target the life stages of Plasmodium parasites that reside within vertebrate red blood cells, as these are the stages that tend to cause disease. However, drugs targeting other stages of the parasite life cycle are under development in order to prevent infection in travelers and to prevent transmission of sexual stages to insect hosts.
File:Saving_Lives_with_SMS_for_Life.jpg|A clinic for treating human malaria in Tanzania File:Anolis carolinensis.jpg|Over 3000 species of lizard, including the Carolina anole (Anolis carolinensis), carry some 90 kinds of malaria.
Insects
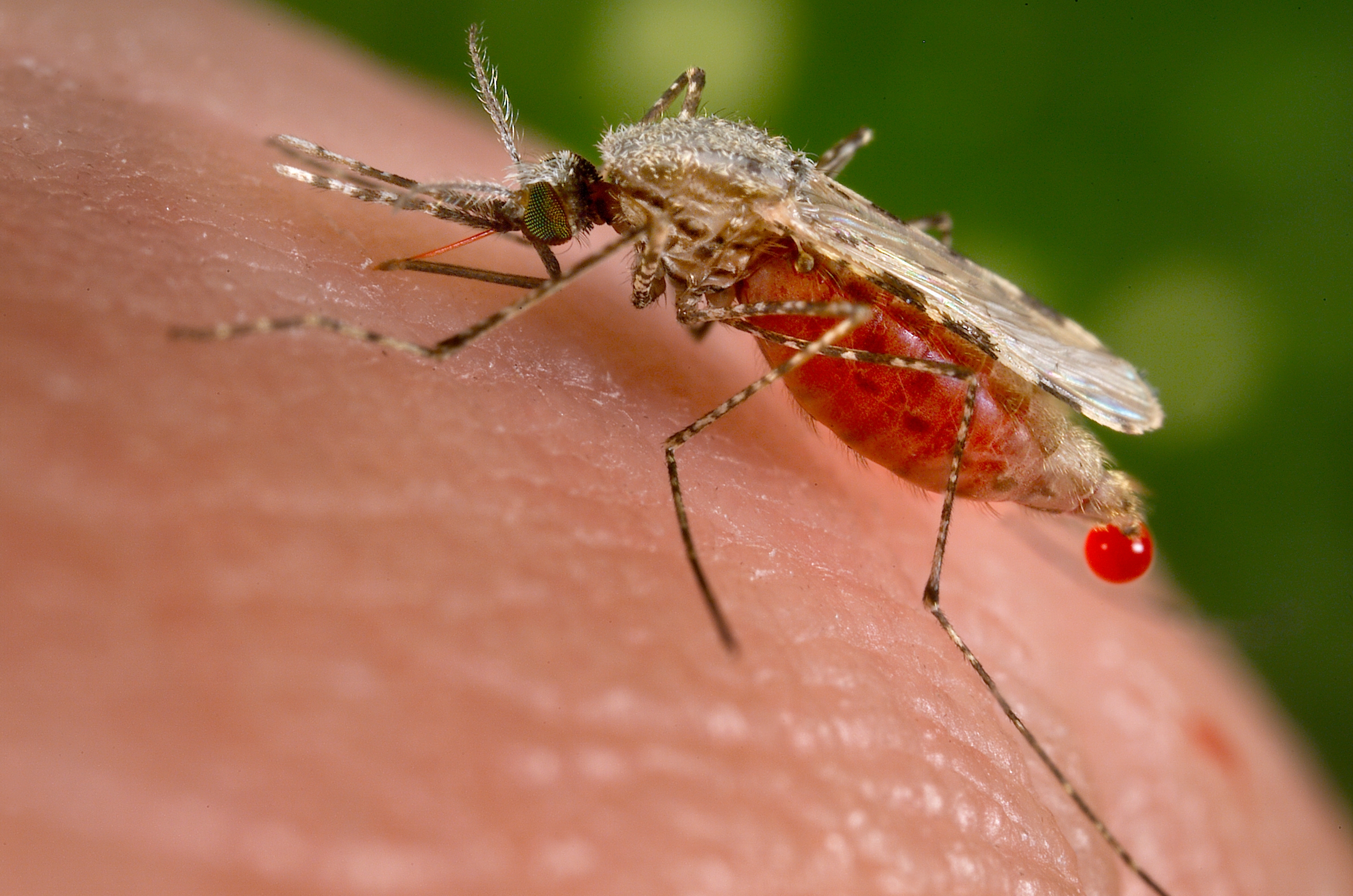
In addition to a vertebrate host, all Plasmodium species also infect a bloodsucking insect host, generally a mosquito (although some reptile-infecting parasites are transmitted by sandflies). Mosquitoes of the genera Culex, Anopheles, Culiseta, Mansonia and Aedes act as insect hosts for various Plasmodium species. The best studied of these are the Anopheles mosquitoes which host the Plasmodium parasites of human malaria, as well as Culex mosquitoes which host the Plasmodium species that cause malaria in birds. Only female mosquitoes are infected with Plasmodium, since only they feed on the blood of vertebrate hosts. Different species affect their insect hosts differently. Sometimes, insects infected with Plasmodium have reduced lifespan and reduced ability to produce offspring. Further, some species of Plasmodium appear to cause insects to prefer to bite infected vertebrate hosts over non-infected hosts.
History
Charles Louis Alphonse Laveran first described parasites in the blood of malaria patients in 1880. He named the parasite Oscillaria malariae. The fact that several species may be involved in causing different forms of malaria was first recognized by Camillo Golgi in 1886. Soon thereafter, Giovanni Batista Grassi and Raimondo Filetti named the parasites causing two different types of human malaria Plasmodium vivax and Plasmodium malariae. In 1897, William Welch identified and named Plasmodium falciparum. This was followed by the recognition of the other two species of Plasmodium which infect humans: Plasmodium ovale (1922) and Plasmodium knowlesi (identified in long-tailed macaques in 1931; in humans in 1965). The contribution of insect hosts to the Plasmodium life cycle was described in 1897 by Ronald Ross and in 1899 by Giovanni Batista Grassi, Amico Bignami and Giuseppe Bastianelli.
In 1966, Cyril Garnham proposed separating Plasmodium into nine subgenera based on host specificity and parasite morphology.
Notes
References
References
- "''Plasmodium''". National Center for Biotechnology Information.
- "CDC – Malaria Parasites – About". U.S. Centers for Disease Control and Prevention.
- (2016). "The nuclear envelope and gene organization in parasitic protozoa: Specializations associated with disease". Molecular and Biochemical Parasitology.
- (2016). "Vacuolar protein sorting mechanisms in apicomplexan parasites". Molecular and Biochemical Parasitology.
- (2013). "Plasmodium rhoptry proteins: Why order is important". Trends in Parasitology.
- (2013). "Subversion of host cellular functions by the apicomplexan parasites". FEMS Microbiology Reviews.
- (2013). "The metabolic roles of the endosymbiotic organelles of Toxoplasma and Plasmodium spp". Current Opinion in Microbiology.
- (2017). "The apicoplast: Now you see it, now you don't". International Journal for Parasitology.
- (June 26, 2013). "The Algal Past and Parasite Present of the Apicoplast". Annual Review of Microbiology.
- (2005). "Malaria: Drugs, Disease, and Post-genomic Biology". Springer.
- "CDC – Malaria Parasites – Biology". U.S. Centers for Disease Control and Prevention.
- Markus, M. B.. (2011). "Malaria: Origin of the Term 'Hypnozoite'". Journal of the History of Biology.
- (2017). "Malaria Parasite Liver Infection and Exoerythrocytic Biology". Cold Spring Harbor Perspectives in Medicine.
- (2022). "Theoretical origin of genetically homologous Plasmodium vivax malarial recurrences". Southern African Journal of Infectious Diseases.
- (2009). "Evolution of the Apicomplexa: Where are we now?". Trends in Parasitology.
- Votypka J. "Haemospororida Danielewski 1885".
- (2013). "Malaria Parasites: Comparative Genomics, Evolution and Molecular Biology". Caister Academic Press.
- Perkins, S. L.. (2014). "Malaria's Many Mates: Past, Present, and Future of the Systematics of the Order Haemosporida". Journal of Parasitology.
- Valkiunas, Gediminas. (2004). "Avian Malaria Parasites and Other Haemosporidia". CRC Press.
- "''Plasmodium''". Tree of Life Web Project.
- (2003). "Progress in Malaria Research: the Case for Phylogenetics".
- (April 2008). "A three-genome phylogeny of malaria parasites (''Plasmodium'' and closely related genera): Evolution of life-history traits and host switches". Molecular Phylogenetics and Evolution.
- (2017). "The geography of imported malaria to non-endemic countries: a meta-analysis of nationally reported statistics". Lancet Infect Dis.
- (2008). "Biodiversity of Malaria in the world". John Libbey.
- (2017). "Molecular interactions governing host-specificity of blood stage malaria parasites". Current Opinion in Microbiology.
- (2006). "Infectious Diseases in Primates: Behavior, Ecology and Evolution". Oxford University Press.
- (2016). "The rediscovery of malaria parasites of ungulates". Parasitology.
- Valkiunas, Gediminas. (2004). "Avian Malaria Parasites and Other Haemosporidia". CRC Press.
- Valkiunas, Gediminas. (2004). "Avian Malaria Parasites and Other Haemosporidia". CRC Press.
- Valkiunas, Gediminas. (2004). "Avian Malaria Parasites and Other Haemosporidia". CRC Press.
- (2012). "Herpetology: An Introductory Biology of Amphibians and Reptiles". Academic Press.
- (2017). "Antimalarial drug resistance: Linking Plasmodium falciparum parasite biology to the clinic". Nature Medicine.
- (2016). "Malaria: Biology and Disease". Cell.
- (2018). "Drug resistance in Plasmodium". Nature Reviews Microbiology.
- (2018). "Multistage inhibitors of the malaria parasite: Emerging hope for chemoprotection and malaria eradication". Medicinal Research Reviews.
- (2014). "Malaria Immunity in Man and Mosquito: Insights into Unsolved Mysteries of a Deadly Infectious Disease". Annual Review of Immunology.
- (2017). "Mechanisms of Plasmodium -Enhanced Attraction of Mosquito Vectors". Trends in Parasitology.
- (2017). "Effects of malaria infection on mosquito olfaction and behavior: Extrapolating data to the field". Current Opinion in Insect Science.
- (2017). "Anopheline Reproductive Biology: Impacts on Vectorial Capacity and Potential Avenues for Malaria Control". Cold Spring Harbor Perspectives in Medicine.
- "The History of Malaria, an Ancient Disease". U.S. Centers for Disease Control and Prevention.
- (2012). "Plasmodia – don't". Trends Parasitol.
- (1963). "New classification of the avian malaria parasites". Parassitologia.
- Telford S. (1988). "A contribution to the systematics of the reptilian malaria parasites, family Plasmodiidae (Apicomplexa: Haemosporina)". Bulletin of the Florida State Museum Biological Sciences.
This article was imported from Wikipedia and is available under the Creative Commons Attribution-ShareAlike 4.0 License. Content has been adapted to SurfDoc format. Original contributors can be found on the article history page.
Ask Mako anything about Plasmodium — get instant answers, deeper analysis, and related topics.
Research with MakoFree with your Surf account
Create a free account to save articles, ask Mako questions, and organize your research.
Sign up freeThis content may have been generated or modified by AI. CloudSurf Software LLC is not responsible for the accuracy, completeness, or reliability of AI-generated content. Always verify important information from primary sources.
Report