From Surf Wiki (app.surf) — the open knowledge base
Pinoresinol
- 4-[(3S,3aR,6S,6aR)-6-(4-hydroxy-3-methoxyphenyl)-1,3,3a,4,6,6a-hexahydrofuro[3,4-c]furan-3-yl]-2-methoxyphenol
- (7α,7′α,8α,8′α)-3,3′-dimethoxy-7,9′:7′,9-diepoxylignane-4,4′-diol (-)-Pinoresinol Pinoresinol is a tetrahydrofuran lignan found in Styrax sp., Forsythia suspensa, and in Forsythia koreana. It is also found in the caterpillar of the cabbage butterfly, Pieris rapae where it serves as a defence against ants.
In food, it is found in sesame seed, in Brassica vegetables and in olive oil. Pinoresinol has also been found to be toxic to larvae of the milkweed bug Oncopeltus fasciatus and of the haematophagous insect Rhodnius prolixus, which is a vector of chagas disease.
Currently, pinoresinol is isolated from plants with low efficiency and low yield.
Biosynthesis
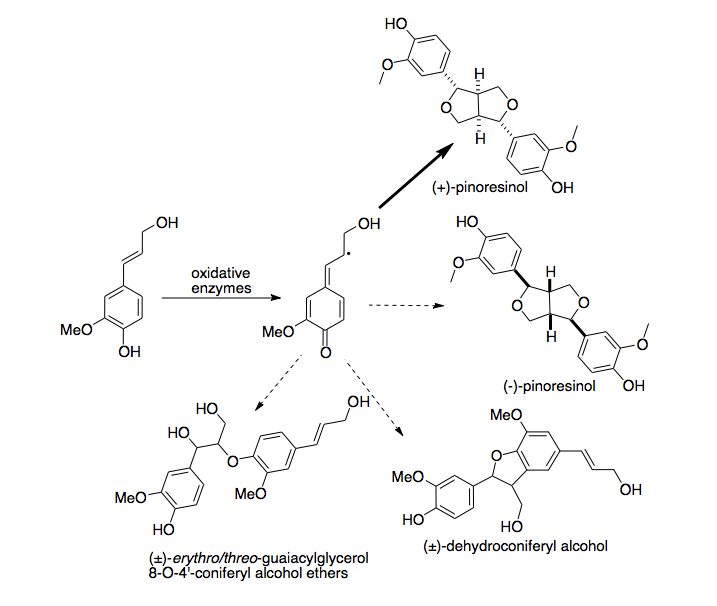
A first dirigent protein was discovered in Forsythia intermedia. This protein has been found to direct the stereoselective biosynthesis of (+)-pinoresinol from coniferyl alcohol monomers. Recently, a second, enantiocomplementary dirigent protein was identified in Arabidopsis thaliana, which directs enantioselective synthesis of (-)-pinoresinol.
Pharmacology
Pinoresinol inhibits the enzyme α-glucosidase in vitro and may therefore act as a hypoglycemic agent. A study involving extra virgin olive oil showed that pinoresinol possess in vitro chemoprevention properties. Increased apoptosis and cellular arrest at the G2/M stage in p53-proficient cells occurred. Pinoresinol of olive oil decreases vitamin D intestinal absorption.
Metabolism into enterolignans
Pinoresinol, along with other plant lignans, are converted into enterolignans by intestinal microflora in the human body.
References
References
- (January 2002). "Pinoresinol from Ipomoea Cairica Cell Cultures". Natural Product Letters.
- Pastrorova ''et al.'' (1997){{full citation needed. (October 2013)
- (August 2010). "Pinoresinol from the fruits of Forsythia koreana inhibits inflammatory responses in LPS-activated microglia". Neuroscience Letters.
- (1992). "On the stereoselective synthesis of (+)-pinoresinol in Forsythia suspensa from its achiral precursor, coniferyl alcohol". Phytochemistry.
- (2006). "Pinoresinol: A lignol of plant origin serving for defense in a caterpillar". Proceedings of the National Academy of Sciences.
- (2007). "Lignan contents of Dutch plant foods: A database including lariciresinol, pinoresinol, secoisolariciresinol and matairesinol". British Journal of Nutrition.
- (2000). "The antioxidant/anticancer potential of phenolic compounds isolated from olive oil". European Journal of Cancer.
- (December 1999). "Effects of the lignan, pinoresinol on the moulting cycle of the bloodsucking bug Rhodnius prolixus and of the milkweed bug Oncopeltus fasciatus". Fitoterapia.
- (2017-05-12). "Engineering of an H2 O2 auto-scavenging in vivo cascade for pinoresinol production". Biotechnology and Bioengineering.
- (1997). "Stereoselective bimolecular phenoxy radical coupling by an auxiliary (dirigent) protein without an active center". Science.
- (March 2007). "An Enantiocomplementary Dirigent Protein for the Enantioselective Laccase-Catalyzed Oxidative Coupling of Phenols". Angewandte Chemie.
- (2012). "(+)-Pinoresinol is a putative hypoglycemic agent in defatted sesame (Sesamum indicum) seeds though inhibiting α-glucosidase". Bioorganic & Medicinal Chemistry Letters.
- (2008). "Chemopreventive properties of pinoresinol-rich olive oil involve a selective activation of the ATM-p53 cascade in colon cancer cell lines". Carcinogenesis.
- (September 2016). "Pinoresinol of olive oil decreases vitamin D intestinal absorption". Food Chemistry.
- (2005). "Lignan contents of Dutch plant foods: A database including lariciresinol, pinoresinol, secoisolariciresinol and matairesinol". The British Journal of Nutrition.
This article was imported from Wikipedia and is available under the Creative Commons Attribution-ShareAlike 4.0 License. Content has been adapted to SurfDoc format. Original contributors can be found on the article history page.
Ask Mako anything about Pinoresinol — get instant answers, deeper analysis, and related topics.
Research with MakoFree with your Surf account
Create a free account to save articles, ask Mako questions, and organize your research.
Sign up freeThis content may have been generated or modified by AI. CloudSurf Software LLC is not responsible for the accuracy, completeness, or reliability of AI-generated content. Always verify important information from primary sources.
Report