From Surf Wiki (app.surf) — the open knowledge base
PAX6
Protein-coding gene in humans
Protein-coding gene in humans
Paired box protein Pax-6, also known as aniridia type II protein (AN2) or oculorhombin, is a protein that in humans is encoded by the PAX6 gene.
Function
PAX6 is a member of the Pax gene family which is responsible for carrying the genetic information that will encode the Pax-6 protein. It acts as a "master control" gene for the development of eyes and other sensory organs, certain neural and epidermal tissues as well as other homologous structures, usually derived from ectodermal tissues. However, it has been recognized that a suite of genes is necessary for eye development, and therefore the term of "master control" gene may be inaccurate. Pax-6 is expressed as a transcription factor when neural ectoderm receives a combination of weak Sonic hedgehog (SHH) and strong TGF-Beta signaling gradients. Expression is first seen in the forebrain, hindbrain, head ectoderm and spinal cord followed by later expression in midbrain. This transcription factor is most noted for its use in the interspecifically induced expression of ectopic eyes and is of medical importance because heterozygous mutants produce a wide spectrum of ocular defects such as aniridia in humans.
Pax6 serves as a regulator in the coordination and pattern formation required for differentiation and proliferation to successfully take place, ensuring that the processes of neurogenesis and oculogenesis are carried out successfully. As a transcription factor, Pax6 acts at the molecular level in the signaling and formation of the central nervous system. The characteristic paired DNA binding domain of Pax6 utilizes two DNA-binding domains, the paired domain (PD), and the paired-type homeodomain (HD). These domains function separately via utilization by Pax6 to carry out molecular signaling that regulates specific functions of Pax6. An example of this lies in HD's regulatory involvement in the formation of the lens and retina throughout oculogenesis contrasted by the molecular mechanisms of control exhibited on the patterns of neurogenesis in brain development by PD. The HD and PD domains act in close coordination, giving Pax6 its multifunctional nature in directing molecular signaling in formation of the CNS. Although many functions of Pax6 are known, the molecular mechanisms of these functions remain largely unresolved. High-throughput studies uncovered many new target genes of the Pax6 transcription factors during lens development. They include the transcriptional activator BCL9, recently identified, together with Pygo2, to be downstream effectors of Pax6 functions.
Role in Human Fetal Development
During human fetal development, PAX6 functions as a master regulatory transcription factor essential for the formation of several organ systems, particularly the eyes, central nervous system, and pancreas. In the developing human eye, PAX6 controls the differentiation and organization of critical structures including the lens, retina, and cornea, and is indispensable for initiating the genetic pathways required for proper ocular formation.[1] Beyond its role in the eye, PAX6 plays a significant part in the patterning of the central nervous system by regulating the proliferation, migration, and specialization of neural progenitor cells during early brain development.[2]
A notable aspect of PAX6's function in the human fetus is its involvement in the regionalization of the developing brain. It helps establish gradients of gene expression within the embryonic forebrain, guiding the formation of distinct cortical regions responsible for sensory, motor, and cognitive functions after birth.[3] Although much of this understanding comes from experimental knockout studies in animal models, similar mutations in humans are associated with serious developmental abnormalities, including aniridia (absence of the iris) and brain malformations, further confirming PAX6's crucial role in human organ development and fetal patterning.[1][2]
References for Role in Human Fetal Development
Hill, R.E., Favor, J., Hogan, B.L., Ton, C.C., Saunders, G.F., Hanson, I.M., Prosser, J., Jordan, T., Hastie, N.D., & van Heyningen, V. (1991). Mouse small eye results from mutations in a paired-like homeobox-containing gene. Nature, 354(6354), 522–525. https://doi.org/10.1038/354522a0 Grindley, J.C., Davidson, D.R., & Hill, R.E. (1995). The role of Pax-6 in eye and nasal development. Development, 121(5), 1433–1442. https://doi.org/10.1242/dev.121.5.1433 Stoykova, A., Treichel, D., Hallonet, M., & Gruss, P. (2000). Pax6 modulates the dorsoventral patterning of the mammalian telencephalon. The Journal of Neuroscience, 20(21), 8042–8050. https://doi.org/10.1523/JNEUROSCI.20-21-08042.2000
Species distribution

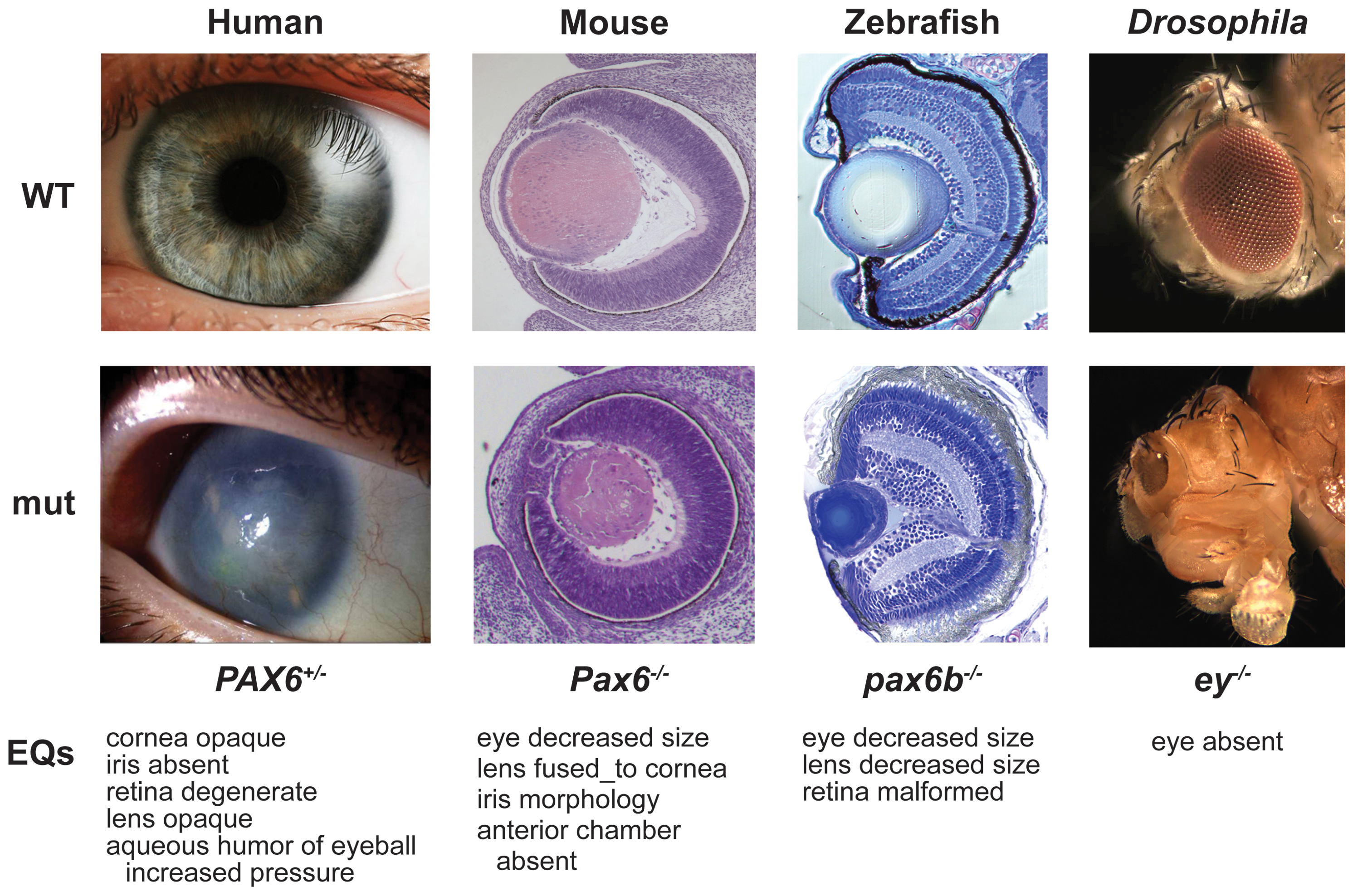
PAX6 protein function is highly conserved across bilaterian species. For instance, mouse PAX6 can trigger eye development in Drosophila melanogaster. Additionally, mouse and human PAX6 have identical amino acid sequences.
Genomic organisation of the PAX6 locus varies among species, including the number and distribution of exons, cis-regulatory elements, and transcription start sites, although most elements at the Vertebrata clade do line up with each other. The first work on genomic organisation was performed in quail, but the picture of the mouse locus is the most complete to date. This consists of 3 confirmed promoters (P0, P1, Pα), 16 exons, and at least 6 enhancers. The 16 confirmed exons are numbered 0 through 13 with the additions of exon α located between exons 4 and 5, and the alternatively spliced exon 5a. Each promoter is associated with its own proximal exon (exon 0 for P0, exon 1 for P1) resulting in transcripts which are alternatively spliced in the 5' un-translated region. By convention, exon for orthologs from other species are named relative to the human/mouse numbering, as long as the organization is reasonably well-conserved.
Of the four Drosophila Pax6 orthologues, it is thought that the eyeless (ey) and twin of eyeless (toy) gene products share functional homology with the vertebrate canonical Pax6 isoform, while the eyegone (eyg) and twin of eyegone (toe) gene products share functional homology with the vertebrate Pax6(5a) isoform. Eyeless and eyegone were named for their respective mutant phenotypes. These paralogs also play a role in the development in the entire eye-antennal disc, and consequently in head formation. toy positively regulates ey expression.
Isoforms
The vertebrate PAX6 locus encodes at least three different protein isoforms, these being the canonical PAX6, PAX6(5a), and PAX6(ΔPD). The canonical PAX6 protein contains an N-terminal paired domain, connected by a linker region to a paired-type homeodomain, and a proline/serine/threonine (P/S/T)-rich C-terminal domain. The paired domain and paired-type homeodomain each have DNA binding activities, while the P/S/T-rich domain possesses a transactivation function. PAX6(5a) is a product of the alternatively spliced exon 5a resulting in a 14 residue insertion in the paired domain which alters the specificity of this DNA binding activity. The nucleotide sequence corresponding to the linker region encodes a set of three alternative translation start codons from which the third PAX6 isoform originates. Collectively known as the PAX6(ΔPD) or pairedless isoforms, these three gene products all lack a paired domain. The pairedless proteins possess molecular weights of 43, 33, or 32kDa, depending on the particular start codon used. PAX6 transactivation function is attributed to the variable length C-terminal P/S/T-rich domain which stretches to 153 residues in human and mouse proteins.
Clinical significance
Experiments in mice demonstrate that a deficiency in Pax-6 leads to decrease in brain size, brain structure abnormality leading to autism, lack of iris formation or a thin cornea. Knockout experiments produced eyeless phenotypes reinforcing indications of the gene's role in eye development. Advancing research in this area may lead us to better understanding of the complexity seen in neural development and maybe one day be able to grow eye tissue in vitro.--
Mutations
During embryological development the PAX6 gene, found on chromosome 2 in mice, can be seen expressed in multiple early structures such as the spinal cord, hindbrain, forebrain and eyes. Mutations of the PAX6 gene in mammalian species can produce a drastic effect on the phenotype of the organism. This can be seen in mice that contain homozygous mutations of the 422 amino acid long transcription factor encoded by PAX6 in which they do not develop eyes or nasal cavities termed 'small eye' mice (PAX10sey/sey). Deletion of PAX6 induces the same abnormal phenotypes indicating that mutations cause the protein to lose functionality. PAX6 is essential is the formation of the retina, lens and cornea due to its role in early cell determination when forming precursors of these structures such as the optic vesicle and overlying surface ectoderm. PAX10 mutations also hinder nasal cavity development due to the similar precursor structures that in small eye mice do not express PAX10 mRNA. Mice lacking any functional pax6 begin to be phenotypically differentiable from normal mouse embryos at about day 9 to 10 of gestation. The full elucidation of the precise mechanisms and molecular components by which the PAX6 gene influences eye, nasal and central nervous system development are still researched however, the study of PAX6 has brought more understanding to the development and genetic complexities of these mammalian body systems.
References
References
- (August 1992). "The human PAX6 gene is mutated in two patients with aniridia". Nature Genetics.
- (2004). "Eyes: variety, development and evolution". Brain, Behavior and Evolution.
- (March 2013). "Functional dissection of the paired domain of Pax6 reveals molecular mechanisms of coordinating neurogenesis and proliferation". Development.
- (August 2015). "Identification of in vivo DNA-binding mechanisms of Pax6 and reconstruction of Pax6-dependent gene regulatory networks during forebrain and lens development". Nucleic Acids Research.
- (September 2014). "Pax6-dependent, but β-catenin-independent, function of Bcl9 proteins in mouse lens development". Genes & Development.
- (September 1999). "Pax 6: mastering eye morphogenesis and eye evolution". Trends in Genetics.
- (May 2008). "Cis-regulatory organization of the Pax6 gene in the ascidian Ciona intestinalis". Developmental Biology.
- (2015). "Pax2/5/8 and Pax6 alternative splicing events in basal chordates and vertebrates: a focus on paired box domain". Frontiers in Genetics.
- (March 2014). "A survey of ancient conserved non-coding elements in the PAX6 locus reveals a landscape of interdigitated cis-regulatory archipelagos". Developmental Biology.
- (2013). "Sequencing of Pax6 loci from the elephant shark reveals a family of Pax6 genes in vertebrate genomes, forged by ancient duplications and divergences". PLOS Genetics.
- (June 2002). "Differential Pax6 promoter activity and transcript expression during forebrain development". Mechanisms of Development.
- (June 2017). "Drosophila Pax6 promotes development of the entire eye-antennal disc, thereby ensuring proper adult head formation". Proceedings of the National Academy of Sciences of the United States of America.
- (August 2004). "Functional divergence between eyeless and twin of eyeless in Drosophila melanogaster". Development.
- (May 2008). "Pax6 3' deletion results in aniridia, autism and mental retardation". Human Genetics.
- (1996). "Transcription factor genes and the developing eye: a genetic perspective". Human Molecular Genetics.
- (December 1991). "Pax-6, a murine paired box gene, is expressed in the developing CNS". Development.
- (May 1995). "The role of Pax-6 in eye and nasal development". Development.
- (June 1995). "Craniofacial abnormalities in homozygous Small eye (Sey/Sey) embryos and newborn mice". Journal of Anatomy.
This article was imported from Wikipedia and is available under the Creative Commons Attribution-ShareAlike 4.0 License. Content has been adapted to SurfDoc format. Original contributors can be found on the article history page.
Ask Mako anything about PAX6 — get instant answers, deeper analysis, and related topics.
Research with MakoFree with your Surf account
Create a free account to save articles, ask Mako questions, and organize your research.
Sign up freeThis content may have been generated or modified by AI. CloudSurf Software LLC is not responsible for the accuracy, completeness, or reliability of AI-generated content. Always verify important information from primary sources.
Report