From Surf Wiki (app.surf) — the open knowledge base
Nierenstein reaction
Chemical reaction
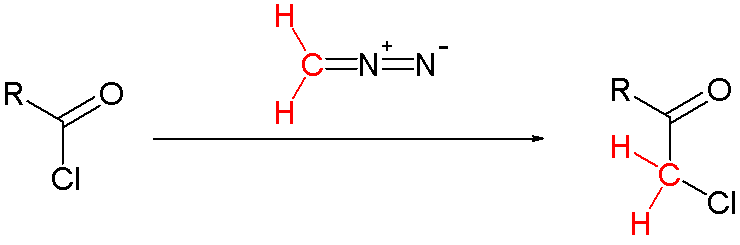
Chemical reaction
The Nierenstein reaction is an organic reaction describing the conversion of an acid chloride into a haloketone with diazomethane.{{cite journal

Reaction mechanism
The reaction proceeds through a diazonium salt intermediate formed when diazomethyl anion displaces the chloride:
Excess diazomethane can act as a base, abstracting a hydrogen from the diazonium intermediate. The results are a neutral diazoketone, which does not react further; and methyldiazonium chloride, which decomposes to chloromethane. The unreactive diazoketone can, however, be re-activated with hydrogen chloride to give the Nierenstein product:
In even some cases with limited diazomethane, the reaction process can stall into the diazoketone pathway, requiring reparative HCl gas. Substitution of a mixed anhydride for the acyl halide also gives the diazoketone.
Examples
Nierenstein's original 1924 publication:{{cite journal

A reaction from benzoyl bromide going haywire and forming the dioxane dimer:{{cite journal

References
References
- McPhee, W. D; Klingsberg, E. ''[[Organic Syntheses]]'', Coll. Vol. 3, p.119 (1955); Vol. 26, p.13 (1946). ([https://orgsyn.org/demo.aspx?prep=cv3p0119 Article])
- Taber, Douglass. (3 July 2017). "The Zhai synthesis of (+)-harringtonolide". Organic Chemistry Highlights.
This article was imported from Wikipedia and is available under the Creative Commons Attribution-ShareAlike 4.0 License. Content has been adapted to SurfDoc format. Original contributors can be found on the article history page.
Ask Mako anything about Nierenstein reaction — get instant answers, deeper analysis, and related topics.
Research with MakoFree with your Surf account
Create a free account to save articles, ask Mako questions, and organize your research.
Sign up freeThis content may have been generated or modified by AI. CloudSurf Software LLC is not responsible for the accuracy, completeness, or reliability of AI-generated content. Always verify important information from primary sources.
Report