From Surf Wiki (app.surf) — the open knowledge base
Gattermann reaction
Chemical reaction
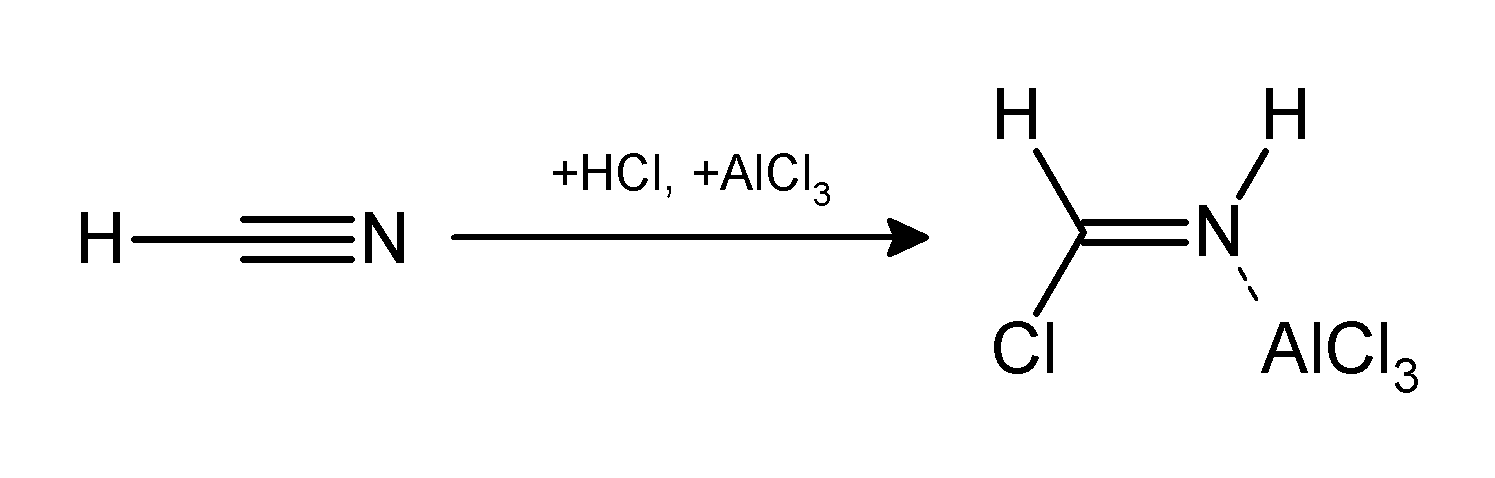
Chemical reaction
The Gattermann reaction (also known as the Gattermann formylation and the Gattermann salicylaldehyde synthesis) is a chemical reaction in which aromatic compounds are formylated by a mixture of hydrogen cyanide (HCN) and hydrogen chloride (HCl) in the presence of a Lewis acid catalyst such as aluminium chloride (AlCl3). It is named for the German chemist Ludwig Gattermann and is similar to the Friedel–Crafts reaction.


Modifications have shown that it is possible to use sodium cyanide or cyanogen bromide in place of hydrogen cyanide.
The reaction can be simplified by replacing the HCN/AlCl3 combination with zinc cyanide. Although it is also highly toxic, Zn(CN)2 is a solid, making it safer to work with than gaseous HCN. The Zn(CN)2 reacts with the HCl to form the key HCN reactant and Zn(Cl)2 that serves as the Lewis-acid catalyst in-situ. An example of the Zn(CN)2 method is the synthesis of mesitaldehyde from mesitylene.
Gattermann–Koch reaction
Julius Arnold Koch The Gattermann–Koch reaction, named after the German chemists Ludwig Gattermann and Julius Arnold Koch, is a variant of the Gattermann reaction in which carbon monoxide (CO) is used instead of hydrogen cyanide.
Unlike the Gattermann reaction, this reaction is not applicable to phenol and phenol ether substrates. Additionally, when zinc chloride is used as the Lewis acid instead of aluminum chloride for example, or when the carbon monoxide is not used at high pressure, the presence of traces of copper(I) chloride or nickel(II) chloride co-catalyst is often necessary. The transition metal co-catalyst may server as a "carrier" by first reacting with CO to form a carbonyl complex, which is then transformed into the active electrophile.
References
ru:Реакция Гаттермана - Коха
References
- {{March6th
- (1898). "Synthese aromatischer Oxyaldehyde". [[Berichte der deutschen chemischen Gesellschaft]].
- (1919). "Über Oxycarbonylverbindungen I. Eine neue Synthese von". Helvetica Chimica Acta.
- (1923). "Simplification of the Gattermann Synthesis of Hydroxy Aldehydes". [[J. Am. Chem. Soc.]].
- (1957). "Organic Reactions, Volume 9". John Wiley & Sons, Inc..
- (1955). "Mesitaldehyde".
- (1897). "Eine Synthese aromatischer Aldehyde". [[Chemische Berichte]].
- (2003). "Name Reactions: A Collection of Detailed Reaction Mechanisms". [[Springer Science+Business Media.
- Kurti, Laszlo.. (2005). "Strategic Applications of Named Reactions in Organic Synthesis : Background and Detailed Mechanisms.". Elsevier Science.
- (1949). "550. The Gattermann–Koch reaction. Part II. Reaction kinetics". J. Chem. Soc..
This article was imported from Wikipedia and is available under the Creative Commons Attribution-ShareAlike 4.0 License. Content has been adapted to SurfDoc format. Original contributors can be found on the article history page.
Ask Mako anything about Gattermann reaction — get instant answers, deeper analysis, and related topics.
Research with MakoFree with your Surf account
Create a free account to save articles, ask Mako questions, and organize your research.
Sign up freeThis content may have been generated or modified by AI. CloudSurf Software LLC is not responsible for the accuracy, completeness, or reliability of AI-generated content. Always verify important information from primary sources.
Report