From Surf Wiki (app.surf) — the open knowledge base
First pass effect
Phenomenon of drug metabolism
Phenomenon of drug metabolism

The first pass effect (FPE), also known as first-pass metabolism (FPM) or presystemic metabolism, is a phenomenon of drug metabolism at a specific location in the body which leads to a reduction in the concentration of the active drug before it reaches the site of action or systemic circulation. The effect is most associated with orally administered medications, but some drugs still undergo first-pass metabolism even when delivered via an alternate route (e.g., IV, IM, etc.). During this metabolism, drug is lost during the process of absorption which is generally related to the liver and gut wall. The liver is the major site of first pass effect; however, it can also occur in the lungs, vasculature or other metabolically active tissues in the body.
Notable drugs that experience a significant first pass effect are buprenorphine, chlorpromazine, cimetidine, diazepam, ethanol (drinking alcohol), imipramine, insulin, lidocaine, midazolam, morphine, pethidine, propranolol, and tetrahydrocannabinol (THC).
First-pass metabolism is not to be confused with phase I metabolism, which is a separate process.
Factors
First-pass metabolism may occur in the liver (for propranolol, lidocaine, clomethiazole, and nitroglycerin) or in the gut (for benzylpenicillin and insulin). The four primary systems that affect the first pass effect of a drug are the enzymes of the gastrointestinal lumen, gastrointestinal wall enzymes, bacterial enzymes and hepatic enzymes.
Hepatic first-pass
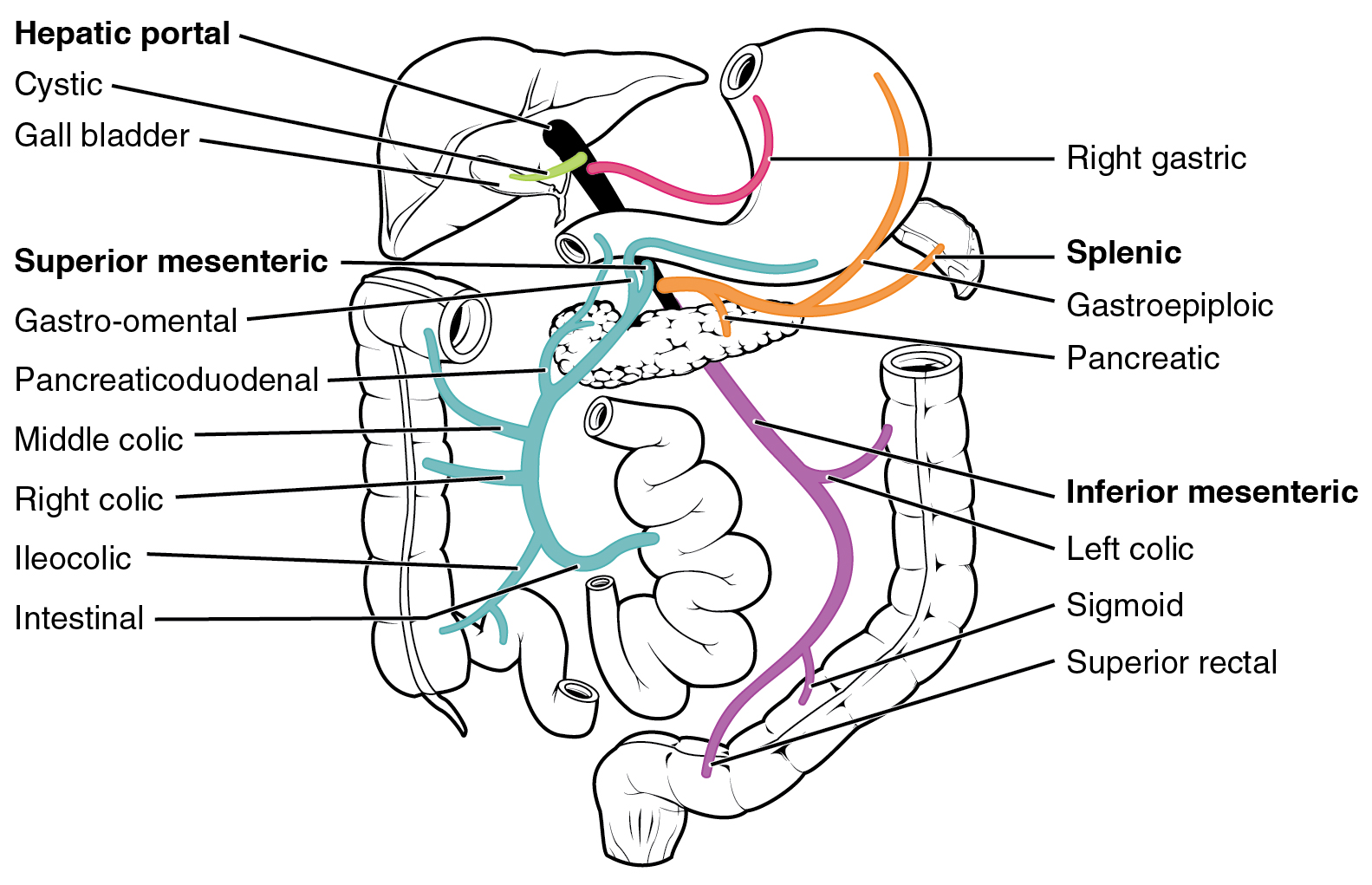
After a water-soluble drug is swallowed, it is absorbed by the digestive system and enters the hepatic portal system. It is carried through the portal vein into the liver before it reaches the rest of the body. The liver metabolizes many drugs, sometimes to such an extent that only a small amount of active drug emerges from the liver to the rest of the circulatory system. This first pass through the liver thus may greatly reduce the bioavailability of the drug.
An example of a drug where first-pass metabolism is a complication and disadvantage is in the antiviral drug remdesivir. Remdesivir cannot be administered orally because the entire dose would be trapped in the liver with little achieving systemic circulation or reaching target organs and cells (for example, cells infected with SARS-CoV-2). For this reason, remdesivir is administered by IV infusion, bypassing the portal vein. However, significant hepatic extraction still occurs because of second pass metabolism, whereby a fraction of venous blood travels through the hepatic portal vein and hepatocytes.
Drug design
In drug design, drug candidates may have good druglikeness but fail on first-pass metabolism because it is biochemically selective. Physiologically based pharmacokinetic models (PBPK) are used to predict first-pass metabolism, although they require compound-specific adjustments due to variability in intestinal mucosal permeability and other factors. Enzyme expression also varies between individuals, which may influence the efficiency of first-pass metabolism and thus the bioavailability of the drug.
Cytochromes P450, especially CYP3A4, play a crucial role in first-pass metabolism, affecting the bioavailability of drugs.
Mitigation
Converting a drug into a prodrug can help avoid first-pass metabolism, thereby improving its bioavailability. In vitro models, such as the use of microfluidic chips that simulate the gut and liver, allow first-pass metabolism to be studied more accurately, facilitating the development of drugs with better absorption profiles.
Routes of administration
Alternative routes of administration, such as insufflation, rectal administration, intravenous, intramuscular, inhalational aerosol, transdermal, or sublingual, avoid or partially avoid the first pass effect because they allow drugs to be absorbed directly into the systemic circulation.
Drugs with high first pass effect typically have a considerably higher oral dose than sublingual or parenteral dose. There is marked individual variation in the oral dose due to differences in the extent of first-pass metabolism, frequently among several other factors. Oral bioavailability of many vulnerable drugs appears to be increased in patients with compromised liver function. Bioavailability is also increased if another drug competing for first-pass metabolism enzymes is given concurrently (e.g., propranolol and chlorpromazine).
References
References
- (January 1972). "Influence of route of administration on drug availability". Journal of Pharmaceutical Sciences.
- (January 1984). "First-Pass Elimination". Clinical Pharmacokinetics.
- Carlin, Michelle G.. (2023-01-01). "Pharmacology and Mechanism of Action of Drugs". Elsevier.
- Bath-Hextall, Fiona. (October 16, 2013). "Understanding First Pass Metabolism". University of Nottingham.
- (1990-01-01). "Mebolism of drugs and other xenobiotics in the gut lumen and wall". Pharmacology & Therapeutics.
- (1997-09-15). "Enzyme-catalyzed processes of first-pass hepatic and intestinal drug extraction". Advanced Drug Delivery Reviews.
- (2018). "Protein Abundance of Clinically Relevant Drug-Metabolizing Enzymes in the Human Liver and Intestine: A Comparative Analysis in Paired Tissue Specimens". Clinical Pharmacology & Therapeutics.
- (2002-04-01). "The Mucosa of the Small Intestine". Clinical Pharmacokinetics.
- (1993). "Gastrointestinal and hepatic first-pass elimination of 2',3'-dideoxyinosine in rats". The Journal of Pharmacology and Experimental Therapeutics.
- (2020). "Advantages of the Parent Nucleoside GS-441524 over Remdesivir for Covid-19 Treatment". ACS Medicinal Chemistry Letters.
- "Fact sheet for health care providers Emergency Use Authorization (EUA) of Veklury®(remdesivir)".
- (2025). "PBPK modeling: What is the role of CYP3A4 expression in the gastrointestinal tract to accurately predict first-pass metabolism?". CPT: Pharmacometrics & Systems Pharmacology.
- (2012-09-29). "Application of PBPK modeling to predict human intestinal metabolism of CYP3A substrates – An evaluation and case study using GastroPlus™". European Journal of Pharmaceutical Sciences.
- (2010). "Prediction of Human Intestinal First-Pass Metabolism of 25 CYP3A Substrates from In Vitro Clearance and Permeability Data". Drug Metabolism and Disposition.
- (2016-05-01). "Gut Wall Metabolism. Application of Pre-Clinical Models for the Prediction of Human Drug Absorption and First-Pass Elimination". The AAPS Journal.
- (2018-01-01). "Chapter 8 - First-Pass Metabolism Considerations in Pharmaceutical Product Development". Academic Press.
- (2021). "Recapitulation of First Pass Metabolism Using 3D Printed Microfluidic Chip and Organoid". Cells.
- (2017-11-07). "3D gut-liver chip with a PK model for prediction of first-pass metabolism". Biomedical Microdevices.
- (2017-01-10). "Microfluidic Gut-liver chip for reproducing the first pass metabolism". Biomedical Microdevices.
- (1997-11-10). "Hepatic first-pass effect and controlled drug delivery following rectal administration". Advanced Drug Delivery Reviews.
- (2022). "Aulton's Pharmaceutics: the design and manufacture of medicines". Elsevier Health Sciences.
- (2009). "Non-invasive Systemic Drug Delivery: Developability Considerations for Alternate Routes of Administration". Journal of Pharmaceutical Sciences.
This article was imported from Wikipedia and is available under the Creative Commons Attribution-ShareAlike 4.0 License. Content has been adapted to SurfDoc format. Original contributors can be found on the article history page.
Ask Mako anything about First pass effect — get instant answers, deeper analysis, and related topics.
Research with MakoFree with your Surf account
Create a free account to save articles, ask Mako questions, and organize your research.
Sign up freeThis content may have been generated or modified by AI. CloudSurf Software LLC is not responsible for the accuracy, completeness, or reliability of AI-generated content. Always verify important information from primary sources.
Report