From Surf Wiki (app.surf) — the open knowledge base
Farinamycin
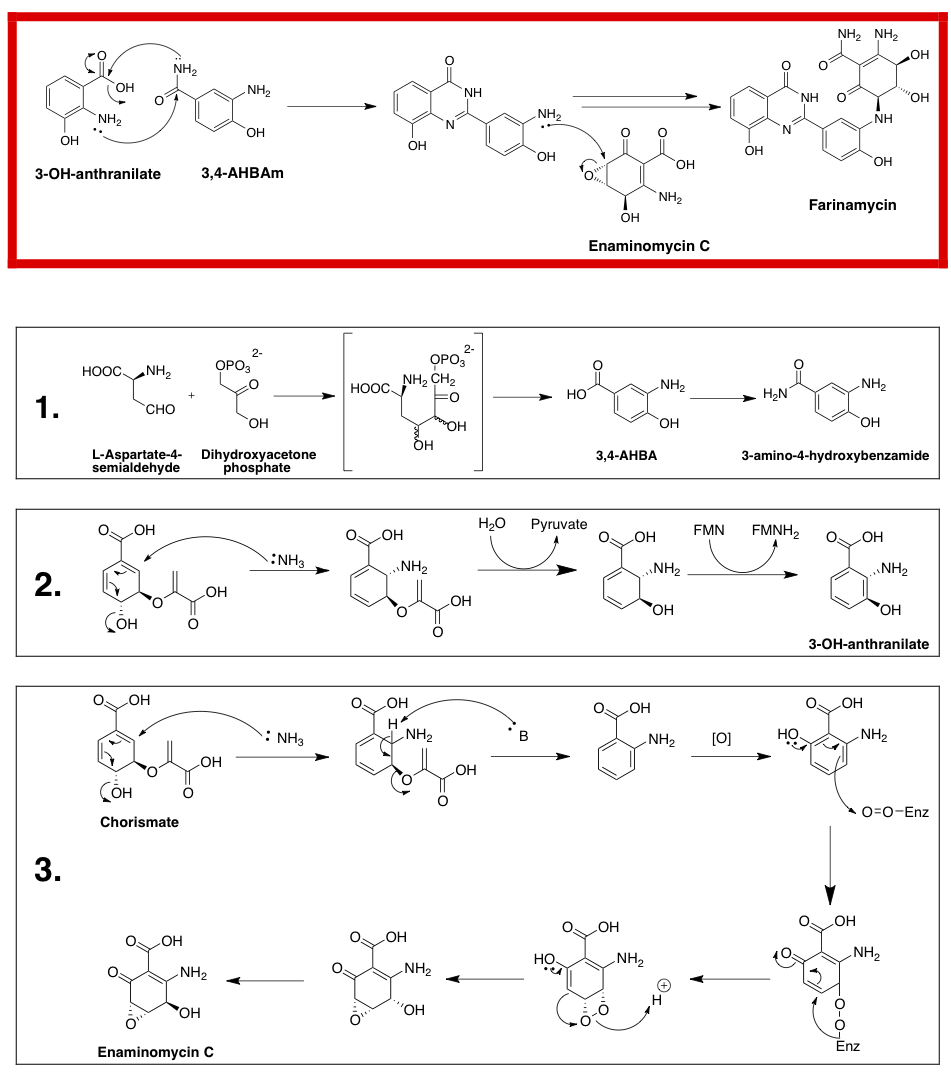
Farinamycin is a quinazoline metabolite that has been isolated from Streptomyces griseus. It is the first known metabolite to be produced by S. griseus that is not a phenoxazinone antibiotic. Farinamycin is formed from the condensation of 3-OH-anthranalite and 3,4-AHBA building blocks that later combine with Enaminomycin C biosynthetically. Many Streptomyces natural products have been used as antibiotics, antifungals, anticancer agents and immunosuppressive agents.
Biosynthesis
Farinamycin is made of three different components: 3-OH-anthranilate (3-HAA), 3,4-aminohydroxybenzamide (3,4-AHBAm) and the natural product Enaminomycin C. 3-OH anthranilate is derived from the central shikimate pathway metabolite chorismate. Chorismate is made from shikimate and is then attacked by nascent ammonia to produce the ortho aminobenzoate isomer by anthranilate synthase. Hydrolysis and FMN mediated reduction leads to the 3-hydroxy aminobenzoate derivative.
3,4-AHBAm is made via the aminoshikimate pathway from L-Aspartate-4-semialdehyde and Dihydroxyacetone phosphate via an enzyme catalyzed aldol condensation followed by ring formation. Enaminomycin C is formed from anthranilate, which is made via the shikimate pathway as well. Subsequent oxidation is followed by a proposed dioxygenase mechanism that leads to epoxide formation. The final step is racemization.
The final quinazoline motif comes from a niementowski-type reaction from 3-HAA and 3,4-AHBAm followed by the nucleophilic oxirane ring opening of enaminomycin C to yield farinamycin.

References
References
- (2011). "Farinamycin, a Qunazoline from "Streptomyces" "griseus"". J. Nat. Prod..
- (2012). "Aminobenzoates as building blocks for natural product assembly lines". Nat. Prod. Rep..
- (2010). "A copper-containing oxidase catalyzes C-nitrosation in nitrosobenzamide biosynthesis". Nature Chemical Biology.
- (1998). "The manumycin-group metabolites". Nat. Prod. Rep..
This article was imported from Wikipedia and is available under the Creative Commons Attribution-ShareAlike 4.0 License. Content has been adapted to SurfDoc format. Original contributors can be found on the article history page.
Ask Mako anything about Farinamycin — get instant answers, deeper analysis, and related topics.
Research with MakoFree with your Surf account
Create a free account to save articles, ask Mako questions, and organize your research.
Sign up freeThis content may have been generated or modified by AI. CloudSurf Software LLC is not responsible for the accuracy, completeness, or reliability of AI-generated content. Always verify important information from primary sources.
Report