From Surf Wiki (app.surf) — the open knowledge base
Dual oxidase 2
Protein-coding gene in the species Homo sapiens
Protein-coding gene in the species Homo sapiens
Dual oxidase 2, also known as DUOX2 or ThOX2 (for thyroid oxidase), is an enzyme that in humans is encoded by the DUOX2 gene. Dual oxidase is an enzyme that was first identified in the mammalian thyroid gland. In humans, two isoforms are found; hDUOX1 and hDUOX2 (this enzyme). The protein location is not exclusive to thyroid tissue; hDUOX1 is prominent in airway epithelial cells and hDUOX2 in the salivary glands and gastrointestinal tract.
Function
Investigations into reactive oxygen species (ROS) in biological systems have, until recently, focused on characterization of phagocytic cell processes. It is now well accepted that production of such species is not restricted to phagocytic cells and can occur in eukaryotic non-phagocytic cell types via NADPH oxidase (NOX) or dual oxidase (DUOX). This new family of proteins, termed the NOX/DUOX family or NOX family of NADPH oxidases, consists of homologs to the catalytic moiety of phagocytic NADPH-oxidase, gp91phox. Members of the NOX/DUOX family have been found throughout eukaryotic species, including invertebrates, insects, nematodes, fungi, amoeba, algae, and plants (not found in prokaryotes). These enzymes clearly demonstrate regulated production of ROS as their sole function. Genetic analyses have implicated NOX/DUOX derived ROS in biological roles and pathological conditions including hypertension (NOX1), innate immunity (NOX2/DUOX), otoconia formation in the inner ear (NOX3) and thyroid hormone biosynthesis (DUOX1/2).DUOX2 is the isoform that generates H2O2 utilized by thyroid peroxidase (TPO) for the biosynthesis of thyroid hormones,Visser, T. J. (2018). Regulation of Thyroid Function, Synthesis, and Function of Thyroid Hormones. In P. Vitti & L. Hegedüs (Eds.), Thyroid Diseases Pathogenesis, Diagnosis, and Treatment. Springer. supported by the discovery of congenital hypothyroidism resultant from an inactivating mutation in the DUOX2 gene.
The family currently has seven members including NOX1, NOX2 (formerly known as gp91phox), NOX3, NOX4, NOX5, DUOX1 and DUOX2.
This protein is known as a dual oxidase because it has both a peroxidase homology domain and a gp91phox domain.
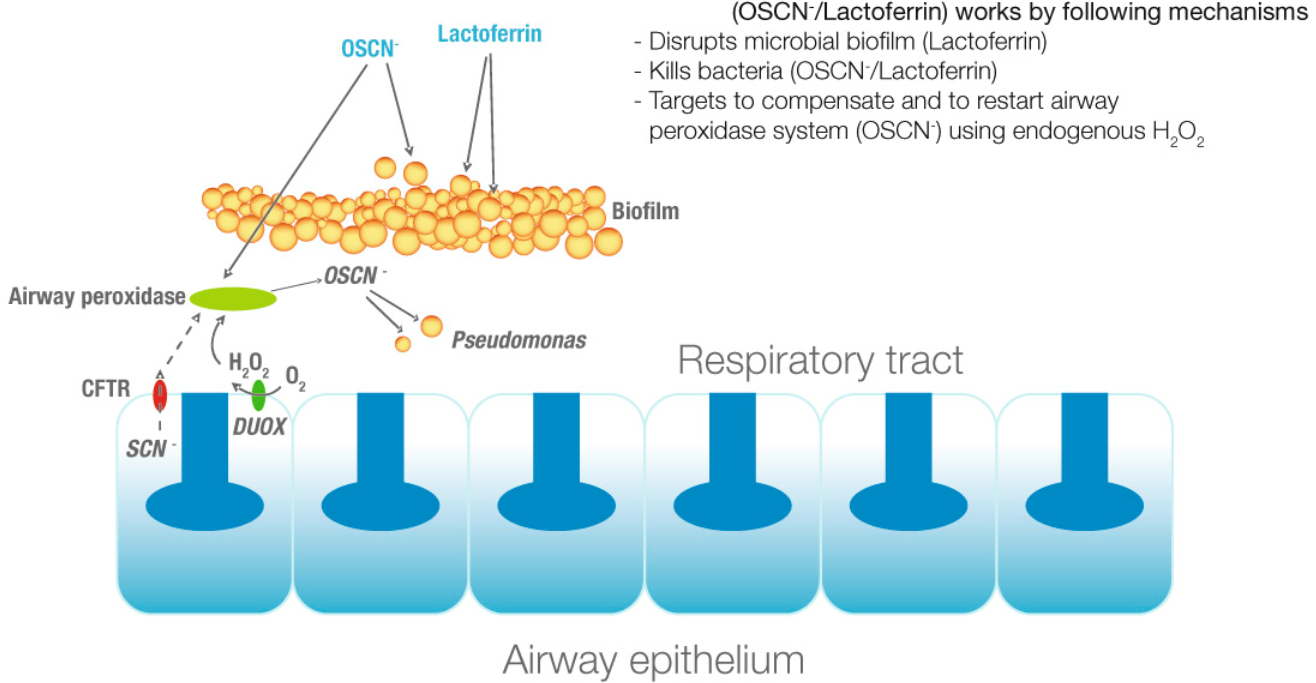
Duox are also implicated in lung defence system and especially in cystic fibrosis.
Schema of duox implication in human lung defence system

References
References
- (December 1999). "Purification of a novel flavoprotein involved in the thyroid NADPH oxidase. Cloning of the porcine and human cdnas". J. Biol. Chem..
- (July 2000). "Cloning of two human thyroid cDNAs encoding new members of the NADPH oxidase family". J. Biol. Chem..
- (August 2005). "Differential regulation of dual NADPH oxidases/peroxidases, Duox1 and Duox2, by Th1 and Th2 cytokines in respiratory tract epithelium". FEBS Lett..
- (August 2003). "Dual oxidases represent novel hydrogen peroxide sources supporting mucosal surface host defense". FASEB J..
- (May 2005). "Dual oxidase2 is expressed all along the digestive tract". Am. J. Physiol. Gastrointest. Liver Physiol..
- (July 2008). "Nox enzymes and oxidative stress in the immunopathology of the gastrointestinal tract". Semin Immunopathol.
- (July 2002). "Inactivating mutations in the gene for thyroid oxidase 2 (THOX2) and congenital hypothyroidism". N. Engl. J. Med..
- "Entrez Gene: DUOX2 dual oxidase 2".
- (October 2009). "Mechanisms and function of DUOX in epithelia of the lung". Antioxid. Redox Signal..
- (October 2008). "The Pseudomonas toxin pyocyanin inhibits the dual oxidase-based antimicrobial system as it imposes oxidative stress on airway epithelial cells". J. Immunol..
- (December 2002). "Lactoperoxidase and hydrogen peroxide metabolism in the airway". Am. J. Respir. Crit. Care Med..
- (2008). "Trends in Innate Immunity". Contrib Microbiol.
This article was imported from Wikipedia and is available under the Creative Commons Attribution-ShareAlike 4.0 License. Content has been adapted to SurfDoc format. Original contributors can be found on the article history page.
Ask Mako anything about Dual oxidase 2 — get instant answers, deeper analysis, and related topics.
Research with MakoFree with your Surf account
Create a free account to save articles, ask Mako questions, and organize your research.
Sign up freeThis content may have been generated or modified by AI. CloudSurf Software LLC is not responsible for the accuracy, completeness, or reliability of AI-generated content. Always verify important information from primary sources.
Report