From Surf Wiki (app.surf) — the open knowledge base
Directed ortho metalation
Chemical reaction
Chemical reaction
Organolithium chemistry
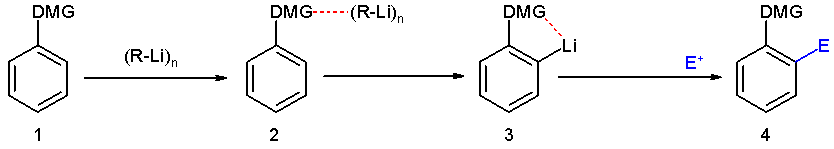
Directed ortho metalation (DoM) is an adaptation of electrophilic aromatic substitution in which electrophiles attach themselves exclusively to the ortho- position of a direct metalation group or DMG through the intermediary of an aryllithium compound. The DMG interacts with lithium through a hetero atom. Examples of DMG's are the methoxy group, a tertiary amine group and an amide group. The compound can be produced by directed lithiation of anisole.

The general principle is outlined in scheme 1. An aromatic ring system with a DMG group 1 interacts with an alkyllithium such as n-butyllithium in its specific aggregation state (hence (R-Li)n) to intermediate 2 since the hetero atom on the DMG is a Lewis base and lithium the Lewis acid. The very basic alkyllithium then deprotonates the ring in the nearest ortho- position forming the aryllithium 3 all the while maintaining the acid-base interaction. An electrophile reacts in the next phase in an electrophilic aromatic substitution with a strong preference for the lithium ipso position replacing the lithium atom.
Ordinary electrophilic substitutions with an activating group show preference for both the ortho and para position, this reaction demonstrates increased regioselectivity because the ortho position alone is targeted.
This reaction type was reported independently by Henry Gilman and Georg Wittig around 1940.
Examples
DOM has traditionally been applied to tertiary anilines and benzyl amines.
The method has also been applied to the synthesis of enantiopure benzyl amines in scheme 3, which involves ortho-lithiation of tert-butyl phenyl sulfoxide. On approach to the lithium intermediate, the bulky tosyl group on the imine electrophile is responsible for the asymmetric induction taking place.

In another application DOM is applied in placing a bulky tert-butyl group in an ortho position (scheme 4). The lithiation is a nucleophilic aromatic substitution and the subsequent reaction to the sulfoxide an electrophilic aromatic substitution. In the final step tert-butyllithium acts as a nucleophile in another nucleophilic aromatic substitution through an anionic intermediate.

DoM has also been applied combined with a Suzuki reaction in a one-pot synthesis:

Thiophenol derivatives
DOM has also been used with thiophenols to prepare compounds that are useful as hindered ligands.
References
References
- (September 1990). "Directed ortho metalation. Tertiary amide and O-carbamate directors in synthetic strategies for polysubstituted aromatics". Chemical Reviews.
- (1979). "Organic Reactions".
- ''Relative Reactivities of Organometallic Compounds. XX.* Metalation'' [[Henry Gilman]], Robert L. Bebb [[J. Am. Chem. Soc.]]; '''1939'''; 61(1); 109-112. {{doi. 10.1021/ja01870a037
- [[Georg Wittig. G. Wittig]] et al. [[Chem. Ber.]] '''1940''', 73, 1197
- (2015). "Directed lithiation of simple aromatics and heterocycles for synthesis of substituted derivatives". Arkivoc.
- J. V. Hay And T. M. Harris "Dimethylamino-5-methylphenyl)diphenylcarbinol" Org. Synth. 1973, volume 53, 56. {{doi. 10.15227/orgsyn.053.0056
- ''ortho-Metalation of Enantiopure Aromatic Sulfoxides and Stereocontrolled Addition to Imines'' Nicolas Le Fur, Ljubica Mojovic, Nelly Plé, Alain Turck, Vincent Reboul, and Patrick Metzner [[J. Org. Chem.]]; '''2006'''; 71(7) pp 2609 - 2616; [https://dx.doi.org/10.1021/jo052358p Abstract]
- Scheme 3. Reaction scheme: reaction of [[iodobenzene]] with [[N-Butyllithium. ''n''-butyllithium]] and (S)-tert-butyl tert-butanethiosulfinate to enantiopure an [[sulfoxide]] followed by DOM reaction initiated again by n-butyllithium with [[electrophile. electrophilic]] N-tosylimine. The sulfoxide group is removed by [[hydrogenation]] with [[Raney nickel]]. ts is a [[tosyl]] group, ee stands for [[enantiomeric excess]]
- ''Contra-Friedel–Crafts tert-butylation of substituted aromatic rings via directed metallation and sulfinylation'' Jonathan Clayden, Christopher C. Stimson and Martine Keenan [[Chemical Communications]], '''2006''', 1393 - 1394 [https://dx.doi.org/10.1039/b600181e Abstract]
- Directed ortho Metalation-Boronation and Suzuki-Miyaura Cross Coupling of Pyridine Derivatives: A One-Pot Protocol to Substituted Azabiaryls Manlio Alessi, Andrew L. Larkin, Kevin A. Ogilvie, Laine A. Green, Sunny Lai, Simon Lopez, and Victor Snieckus [[J. Org. Chem.]]; '''2007'''; 72(5) pp 1588 - 1594. {{doi. 10.1021/jo0620359
- In this sequence the starting material [[nicotinamide]] is lithiated, then reacted with triisopropoxyborane to a [[boronate ester]], then reacted with [[pinacol]] and finally reacted with [[iodobenzene]] and [[Tetrakis(triphenylphosphine)palladium(0)]]
- Directed ortho-lithiation of lithium thiophenolate. New methodology for the preparation of ortho-substituted thiophenols and related compounds Garret D. Figuly, Cynthia K. Loop, J. C. Martin [[J. Am. Chem. Soc.]]; '''1989'''; 111 pp 654-658 {{doi. 10.1021/ja00184a038. Ortho-Lithiothiophenol Equivalents: Generation, Reactions and Applications in Synthesis of Hindered Thiolate Ligands Eric Block, Venkatachalam Eswarakrishnan, Michael Gernon, Gabriel Ofori-Okai, Chantu Saha, Kaluo Tang, Jon Zubieta [[J. Am. Chem. Soc.]]; '''1989'''; 111 pp 658-665. {{doi. 10.1021/ja00184a039. Directed lithiation of arenethiols Keith Smith, Charles M. Lindsay, Gareth J. Pritchard [[J. Am. Chem. Soc.]]; '''1989'''; 111 pp 665-669; {{doi. 10.1021/ja00184a040. 2-Phosphino- and 2-Phosphinyl-benzenethiols: New Ligand Types Eric Block, Gabriel Ofori-Okai and Jon Zubieta [[J. Am. Chem. Soc.]]; '''1989'''; 111 pp 2327-2329; {{doi. 10.1039/B511827A
- Solvent [[hexane]] reaction at [[room temperature]]. Selected bond lengths in 2: Zn-C bond 203.5 pm in plane with aryl plane, Na-C bond 269 pm at 76° to aryl plane
This article was imported from Wikipedia and is available under the Creative Commons Attribution-ShareAlike 4.0 License. Content has been adapted to SurfDoc format. Original contributors can be found on the article history page.
Ask Mako anything about Directed ortho metalation — get instant answers, deeper analysis, and related topics.
Research with MakoFree with your Surf account
Create a free account to save articles, ask Mako questions, and organize your research.
Sign up freeThis content may have been generated or modified by AI. CloudSurf Software LLC is not responsible for the accuracy, completeness, or reliability of AI-generated content. Always verify important information from primary sources.
Report