From Surf Wiki (app.surf) — the open knowledge base
Dextroamphetamine
CNS stimulant and isomer of amphetamine
CNS stimulant and isomer of amphetamine
| Field | Value | |
|---|---|---|
| Verifiedfields | changed | |
| Watchedfields | changed | |
| verifiedrevid | 596899008 | |
| INN | Dexamfetamine | |
| image | D-Amphetamine.svg | |
| image_class | skin-invert-image | |
| width | 210 | |
| imageL | D-Amphetamine-3D-balls.png | |
| image_classL | bg-transparent | |
| imageR | Amphetamine-3d-CPK.png | |
| image_classR | bg-transparent | |
| caption | ||
| pronounce | ||
| tradename | Dexedrine, Zenzedi, others | |
| Drugs.com | ||
| MedlinePlus | a605027 | |
| DailyMedID | Dextroamphetamine | |
| pregnancy_AU | B3 | |
| dependency_liability | Physical: None | |
| Psychological: Moderate | ||
| <!-- PLEASE NOTE: countless sources state that amphetamine has a "High" abuse liability. This term is not synonymous with "addiction liability", which is the relative risk (compared to other addictive drugs) of developing an addiction (aka "substance use disorder") when it's used as prescribed or recreationally. Also note this is the same dependence/addiction breakdown that's on the "Amphetamine" page. Important to distinguish between physical and psychological dependence. --> | addiction_liability | Moderate |
| routes_of_administration | By mouth, transdermal, intravenous, insufflation, rectal | |
| class | Stimulant | |
| ATC_prefix | N06 | |
| ATC_suffix | BA02 | |
| ATC_supplemental | ||
| legal_AU | S8 | |
| legal_AU_comment | ||
| legal_BR | A3 | |
| legal_BR_comment | ||
| legal_CA | Rx-only | |
| legal_CA_comment | /Schedule G (CDSA I) | |
| legal_DE | Anlage III | |
| legal_NZ | ||
| legal_UK | Class B | |
| legal_US | Schedule II | |
| legal_US_comment | ||
| legal_EU | Rx-only | |
| legal_EU_comment | ||
| legal_UN | P II | |
| legal_status | SE: Förteckning II | |
| <!-- Pharmacokinetic data --> | bioavailability | Oral: ~90% |
| protein_bound | 15–40% | |
| metabolism | CYP2D6, DBH, FMO3 | |
| onset | IR dosing: 30–45 minutes | |
| XR dosing: 1.5–2 hours | ||
| elimination_half-life | 9–11 hours | |
| pH-dependent: 7–34 hours | ||
| duration_of_action | IR dosing: 3–6 hours | |
| XR dosing: 8–12 hours | ||
| excretion | Kidney (45%); urinary pH-dependent | |
| <!-- Identifiers --> | CAS_number_Ref | |
| CAS_number | 51-64-9 | |
| PubChem | 5826 | |
| IUPHAR_ligand | 2147 | |
| DrugBank_Ref | ||
| DrugBank | DB01576 | |
| ChemSpiderID_Ref | ||
| ChemSpiderID | 5621 | |
| UNII_Ref | ||
| UNII | TZ47U051FI | |
| KEGG_Ref | ||
| KEGG | D03740 | |
| ChEBI_Ref | ||
| ChEBI | 4469 | |
| ChEMBL_Ref | ||
| ChEMBL | 612 | |
| synonyms | d-Amphetamine, (S)-Amphetamine, S(+)-Amphetamine | |
| <!-- Chemical and physical data --> | IUPAC_name | (2S)-1-Phenylpropan-2-amine |
| C | 9 | |
| H | 13 | |
| N | 1 | |
| chirality | Dextrorotatory enantiomer | |
| SMILES | CC@@HN | |
| StdInChI_Ref | ||
| StdInChI | InChI=1S/C9H13N/c1-8(10)7-9-5-3-2-4-6-9/h2-6,8H,7,10H2,1H3/t8-/m0/s1 | |
| StdInChIKey_Ref | ||
| StdInChIKey | KWTSXDURSIMDCE-QMMMGPOBSA-N | |
| density | 0.913 | |
| boiling_point | 201.5 | |
| solubility | 20 | |
| sol_units | mg per ml |
| Drugs.com = Psychological: Moderate
XR dosing: 1.5–2 hours | elimination_half-life = 9–11 hours pH-dependent: 7–34 hours XR dosing: 8–12 hours
Dextroamphetamine is a central nervous system (CNS) stimulant and enantiomer of amphetamine that is used in the treatment of attention deficit hyperactivity disorder (ADHD) and narcolepsy. It is also used illicitly to enhance cognitive and athletic performance, and recreationally as an aphrodisiac and euphoriant. Dextroamphetamine is generally regarded as the prototypical stimulant.
The amphetamine molecule exists as two enantiomers, levoamphetamine and dextroamphetamine. Dextroamphetamine is the dextrorotatory, or 'right-handed', enantiomer and exhibits more pronounced effects on the central nervous system than levoamphetamine. Pharmaceutical dextroamphetamine sulfate is available as both a brand name and generic drug in a variety of dosage forms. Dextroamphetamine is sometimes prescribed as the inactive prodrug lisdexamfetamine.
Side effects of dextroamphetamine at therapeutic doses include elevated mood, decreased appetite, dry mouth, excessive grinding of the teeth, headache, increased heart rate, increased wakefulness or insomnia, anxiety, and irritability, among others. At excessive doses, psychosis (i.e., hallucinations, delusions), addiction, and rapid muscle breakdown may occur. However, for individuals with pre-existing psychotic disorders, there may be a risk of psychosis even at therapeutic doses.
Dextroamphetamine, like other amphetamines, elicits its stimulating effects via several distinct actions: it inhibits or reverses the transporter proteins for the monoamine neurotransmitters (namely the serotonin, norepinephrine and dopamine transporters) either via trace amine-associated receptor 1 (TAAR1) or in a TAAR1 independent fashion when there are high cytosolic concentrations of the monoamine neurotransmitters In 2022, mixed amphetamine salts (Adderall) was the 14th most commonly prescribed medication in the United States, with more than 34million prescriptions.
Uses
Medical

Dextroamphetamine is used to treat attention deficit hyperactivity disorder (ADHD) and narcolepsy, and is sometimes prescribed off-label for depression and obesity.
ADHD
Narcolepsy
Enhancing performance
Recreational
Dextroamphetamine is also used recreationally as a euphoriant and aphrodisiac, and, like other amphetamines, is used as a club drug for its energetic and euphoric high. Dextroamphetamine is considered to have a high potential for misuse in a recreational manner since individuals typically report feeling euphoric, more alert, and more energetic after taking the drug. Dextroamphetamine's dopaminergic (rewarding) properties affect the mesocorticolimbic circuit; a group of neural structures responsible for incentive salience (i.e., "wanting"; desire or craving for a reward and motivation), positive reinforcement and positively-valenced emotions, particularly ones involving pleasure. Large recreational doses of dextroamphetamine may produce dextroamphetamine overdose. Recreational users sometimes open dexedrine capsules and crush the contents in order to insufflate (snort) it or subsequently dissolve it in water and inject it. Immediate-release formulations have higher potential for abuse via insufflation (snorting) or intravenous injection due to a more favorable pharmacokinetic profile and easy crushability (especially tablets).
The reason for using crushed spansules for insufflation and injection methods is evidently due to the instant-release forms of the drug seen in tablet preparations often containing a sizable amount of inactive binders and fillers alongside the active d-amphetamine, such as dextrose. Injection into the bloodstream can be dangerous because insoluble fillers within the tablets can block small blood vessels. Chronic overuse of dextroamphetamine can lead to severe drug dependence, resulting in withdrawal symptoms when drug use stops.
Contraindications
Adverse effects
Overdose
Interactions
Many types of substances are known to interact with amphetamine, resulting in altered drug action or metabolism of amphetamine, the interacting substance, or both. Inhibitors of the enzymes that metabolize amphetamine (e.g., CYP2D6 and FMO3) will prolong its elimination half-life, meaning that its effects will last longer. Amphetamine also interacts with MAOIs, particularly monoamine oxidase A inhibitors, since both MAOIs and amphetamine increase plasma catecholamines (i.e., norepinephrine and dopamine); therefore, concurrent use of both is dangerous. Amphetamine modulates the activity of most psychoactive drugs. In particular, amphetamine may decrease the effects of sedatives and depressants and increase the effects of stimulants and antidepressants. Amphetamine may also decrease the effects of antihypertensives and antipsychotics due to its effects on blood pressure and dopamine respectively. Zinc supplementation may reduce the minimum effective dose of amphetamine when it is used for the treatment of ADHD. Norepinephrine reuptake inhibitors (NRIs) like atomoxetine prevent norepinephrine release induced by amphetamines and have been found to reduce the stimulant, euphoriant, and sympathomimetic effects of dextroamphetamine in humans.
Pharmacology
Pharmacodynamics
| Compound | Ref |
|---|---|
| Phenethylamine | 10.9 |
| Dextroamphetamine | 6.6–7.2 |
| Levoamphetamine | 9.5 |
| Dextromethamphetamine | 12.3–13.8 |
| Levomethamphetamine | 28.5 |
| Notes: The smaller the value, the more strongly the drug releases the neurotransmitter. See also Monoamine releasing agent § Activity profiles for a larger table with more compounds. Refs: |
Amphetamine and its enantiomers have been identified as potent full agonists of trace amine-associated receptor 1 (TAAR1), a GPCR, discovered in 2001, that is important for regulation of monoaminergic systems in the brain. Activation of TAAR1 increases cAMP production via adenylyl cyclase activation and inhibits the function of the dopamine transporter, norepinephrine transporter, and serotonin transporter, as well as inducing the release of these monoamine neurotransmitters (effluxion). Amphetamine enantiomers are also substrates for a specific neuronal synaptic vesicle uptake transporter called VMAT2. When amphetamine is taken up by VMAT2, the vesicle releases (effluxes) dopamine, norepinephrine, and serotonin, among other monoamines, into the cytosol in exchange.
Dextroamphetamine (the dextrorotary enantiomer) and levoamphetamine (the levorotary enantiomer) have identical pharmacodynamics, but their binding affinities to their biomolecular targets vary. Dextroamphetamine is a more potent agonist of TAAR1 than levoamphetamine. Consequently, dextroamphetamine produces roughly three to four times more central nervous system (CNS) stimulation than levoamphetamine; however, levoamphetamine has slightly greater cardiovascular and peripheral effects.
Related endogenous compounds
Pharmacokinetics
History, society, and culture
Main article: History and culture of amphetamines
Racemic amphetamine was first synthesized under the chemical name "phenylisopropylamine" in Berlin, 1887 by the Romanian chemist Lazăr Edeleanu. It was not widely marketed until 1932, when the pharmaceutical company Smith, Kline & French (now known as GlaxoSmithKline) introduced it in the form of the Benzedrine inhaler for use as a bronchodilator. Notably, the amphetamine contained in the Benzedrine inhaler was the liquid free-base,Free-base form amphetamine is a volatile oil, hence the efficacy of the inhalers. not a chloride or sulfate salt.
In 1935, the medical community became aware of the stimulant properties of amphetamine, specifically the dextroamphetamine isomer, and in 1937 Smith, Kline, and French introduced tablets under the brand name Dexedrine. In the United States, Dexedrine was approved to treat narcolepsy and attention deficit hyperactivity disorder (ADHD). In Canada indications once included epilepsy and parkinsonism. Dextroamphetamine was marketed in various other forms in the following decades, primarily by Smith, Kline, and French, such as several combination medications including a mixture of dextroamphetamine and amobarbital (a barbiturate) sold under the brand name Dexamyl and, in the 1950s, an extended release capsule (the "Spansule"). Preparations containing dextroamphetamine were also used in World War II as a treatment against fatigue.
It quickly became apparent that dextroamphetamine and other amphetamines had a high potential for misuse, although they were not heavily controlled until 1970, when the Comprehensive Drug Abuse Prevention and Control Act was passed by the United States Congress. Dextroamphetamine, along with other sympathomimetics, was eventually classified as Schedule II, the most restrictive category possible for a drug with a government-sanctioned, recognized medical use. Internationally, it has been available under the names AmfeDyn (Italy), Curban (US), Obetrol (Switzerland), Simpamina (Italy), Dexedrine/GSK (US & Canada), Dexedrine/UCB (United Kingdom), Dextropa (Portugal), and Stild (Spain). It became popular on the mod scene in England in the early 1960s, and carried through to the Northern Soul scene in the north of England to the end of the 1970s.
In October 2010, GlaxoSmithKline sold the rights for Dexedrine Spansule to Amedra Pharmaceuticals (a subsidiary of CorePharma).
The U.S. Air Force uses dextroamphetamine as one of its "go pills", given to pilots on long missions to help them remain focused and alert. Conversely, "no-go pills" are used after the mission is completed, to combat the effects of the mission and "go-pills". The Tarnak Farm incident was linked by media reports to the use of this drug on long term fatigued pilots. The military did not accept this explanation, citing the lack of similar incidents. Newer stimulant medications or awakeness promoting agents with different side effect profiles, such as modafinil, are being investigated and sometimes issued for this reason.
Formulations
| Brand | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| name | United States | |||||||||||
| Adopted Name | (D:L) ratio | Dosage | ||||||||||
| form | Marketing | |||||||||||
| start date | Sources | |||||||||||
| Adderall | Mixed amphetamine salts | 3:1 (salts) | tablet | 1996 | ||||||||
| Adderall XR | Mixed amphetamine salts | 3:1 (salts) | capsule | 2001 | title = National Drug Code Amphetamine Search Results | url = http://www.accessdata.fda.gov/scripts/cder/ndc/results.cfm?beginrow=1&numberperpage=160&searchfield=amphetamine&searchtype=ActiveIngredient&OrderBy=ProprietaryName | website = National Drug Code Directory | publisher=U.S. Food and Drug Administration (FDA) | access-date = 16 December 2013 | archive-url = https://web.archive.org/web/20131216080856/http://www.accessdata.fda.gov/scripts/cder/ndc/results.cfm?beginrow=1&numberperpage=160&searchfield=amphetamine&searchtype=ActiveIngredient&OrderBy=ProprietaryName | archive-date=16 December 2013}} | |
| Mydayis | Mixed amphetamine salts | 3:1 (salts) | capsule | 2017 | title=Mydayis- dextroamphetamine sulfate, dextroamphetamine saccharate, amphetamine aspartate monohydrate, and amphetamine sulfate capsule, extended release | website=DailyMed | date=28 October 2022 | url=https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=141a7970-3f06-44ea-9ab7-aeece2c085fc | access-date=21 January 2023}} | |||
| Adzenys XR-ODT | amphetamine | 3:1 (base) | ODT | 2016 | title=Adzenys XR-ODT- amphetamine tablet, orally disintegrating | website=DailyMed | date=10 March 2022 | url=https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=c1179269-00b5-48ea-972d-31e614e99b7e | access-date=21 January 2023}} | |||
| Dyanavel XR | amphetamine | 3.2:1 (base) | suspension | 2015 | title=Drug Approval Package: Dyanavel XR | website=U.S. Food and Drug Administration (FDA) | date=21 March 2022 | url=https://www.accessdata.fda.gov/drugsatfda_docs/nda/2022/210526Orig1s000TOC.cfm | archive-url=https://web.archive.org/web/20221027223300/https://www.accessdata.fda.gov/drugsatfda_docs/nda/2022/210526Orig1s000TOC.cfm | url-status=dead | archive-date=27 October 2022 | access-date=21 January 2023}} |
| Evekeo | amphetamine sulfate | 1:1 (salts) | tablet | 2012 | Contraindications -- | |||||||
| Dexedrine | dextroamphetamine sulfate | 1:0 (salts) | capsule | 1976 | ||||||||
| Zenzedi | dextroamphetamine sulfate | 1:0 (salts) | tablet | 2013 | ||||||||
| Vyvanse | lisdexamfetamine dimesylate | 1:0 (prodrug) | capsule | 2007 | ||||||||
| tablet | ||||||||||||
| Xelstrym | dextroamphetamine | 1:0 (base) | patch | 2022 |
Transdermal dextroamphetamine patches
Dextroamphetamine is available as a transdermal patch containing dextroamphetamine base under the brand name Xelstrym.
Dextroamphetamine sulfate
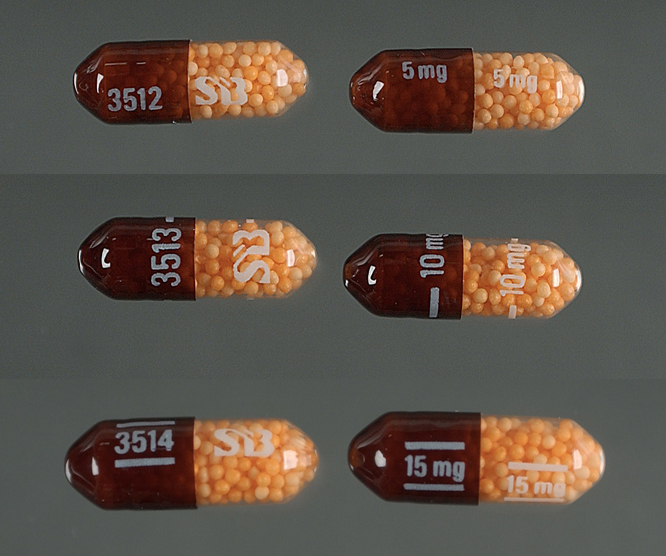
In the United States, immediate release (IR) formulations of dextroamphetamine sulfate are available generically as 5 mg and 10 mg tablets, marketed by Barr (Teva Pharmaceutical Industries), Mallinckrodt Pharmaceuticals, Wilshire Pharmaceuticals, Aurobindo Pharmaceutical USA and CorePharma. Previous IR tablets sold under the brand names Dexedrine and Dextrostat have been discontinued but in 2015, IR tablets became available by the brand name Zenzedi, offered as 2.5 mg, 5 mg, 7.5 mg, 10 mg, 15 mg, 20 mg and 30 mg tablets. Dextroamphetamine sulfate is also available as a controlled-release (CR) capsule preparation in strengths of 5 mg, 10 mg, and 15 mg under the brand name Dexedrine Spansule, with generic versions marketed by Barr and Mallinckrodt. A bubblegum flavored oral solution is available under the brand name ProCentra, manufactured by FSC Pediatrics, which is designed to be an easier method of administration in children who have difficulty swallowing tablets, each 5 mL contains 5 mg dextroamphetamine. The conversion rate between dextroamphetamine sulfate to amphetamine free base is .728.
In Australia, dexamfetamine is available in bottles of 100 instant release 5 mg tablets as a generic drug or slow release dextroamphetamine preparations may be compounded by individual chemists. In the United Kingdom, it is available in 5 mg instant release sulfate tablets under the generic name dexamfetamine sulfate as well as 10 mg and 20 mg strength tablets under the brand name Amfexa. It is also available in generic dexamfetamine sulfate 5 mg/ml oral sugar-free syrup. The brand name Dexedrine was available in the United Kingdom prior to UCB Pharma disinvesting the product to another pharmaceutical company, Auden Mckenzie.
Lisdexamfetamine
Main article: Lisdexamfetamine
Dextroamphetamine is the active metabolite of the prodrug lisdexamfetamine (L-lysine-dextroamphetamine), available by the brand name Vyvanse (Elvanse in the European market and Venvanse in the Brazilian market). Dextroamphetamine is liberated from lisdexamfetamine enzymatically following contact with red blood cells. The conversion is rate-limited by the enzyme, which prevents high blood concentrations of dextroamphetamine and reduces lisdexamfetamine's drug liking and abuse potential at clinical doses. Vyvanse is marketed as once-a-day dosing as it provides a slow release of dextroamphetamine into the body. Vyvanse is available as capsules, and chewable tablets, and in seven strengths; 10 mg, 20 mg, 30 mg, 40 mg, 50 mg, 60 mg, and 70 mg. The conversion rate between lisdexamfetamine dimesylate (Vyvanse) to dextroamphetamine base is 29.5%.
Adderall
Main article: Adderall

Another pharmaceutical that contains dextroamphetamine is commonly known by the brand name Adderall. It is available as immediate release (IR) tablets and extended release (XR) capsules. Adderall contains equal amounts of four amphetamine salts:
- One-quarter racemic (d,l-)amphetamine aspartate monohydrate
- One-quarter dextroamphetamine saccharate
- One-quarter dextroamphetamine sulfate
- One-quarter racemic (d,l-)amphetamine sulfate
Adderall has a total amphetamine base equivalence of 63%. While the enantiomer ratio by dextroamphetamine salts to levoamphetamine salts is 3:1, the amphetamine base content is 75.9% dextroamphetamine, 24.1% levoamphetamine.
Research
Schizophrenia
Dextroamphetamine reduces the negative symptoms of schizophrenia, and has been shown to enhance the effects of auditory discrimination training in schizophrenic patients.
Notes
;Image legend
Reference notes
References
References
- (26 September 2022). "Therapeutic Goods (Poisons Standard—February 2023) Instrument 2022".
- (20 February 2022). "ADHD Stimulant Prescribing Regulations & Authorities in Australia & New Zealand".
- Anvisa. (31 March 2023). "RDC Nº 784 - Listas de Substâncias Entorpecentes, Psicotrópicas, Precursoras e Outras sob Controle Especial". [[Diário Oficial da União]].
- (6 June 2024). "Product monograph brand safety updates".
- (10 January 2022). "Dexedrine spansule- dextroamphetamine sulfate capsule, extended release".
- (6 January 2023). "Xelstrym- dextroamphetamine patch, extended release".
- "List of nationally authorised medicinal products : Active substance(s): dexamfetamine : Procedure No. PSUSA/00000986/202109".
- (2013). "Foye's Principles of Medicinal Chemistry". Wolters Kluwer Health/Lippincott Williams & Wilkins.
- (1 January 2001). "Primary Care Pediatrics". Lippincott Williams & Wilkins.
- (2010). "Attention Deficit Hyperactivity Disorder Handbook: A Physician's Guide to ADHD". Springer.
- (September 2008). "Onset of efficacy of long-acting psychostimulants in pediatric attention-deficit/hyperactivity disorder". Postgrad. Med..
- (27 February 2022). "Adderall- dextroamphetamine saccharate, amphetamine aspartate, dextroamphetamine sulfate, and amphetamine sulfate tablet".
- (October 2012). "A practical guide to the therapy of narcolepsy and hypersomnia syndromes". Neurotherapeutics.
- (August 2006). "dextrostat (dextroamphetamine sulfate) tablet [Shire US Inc.]". Shire US Inc..
- "Dextroamphetamine Monograph for Professionals". American Society of Health-System Pharmacists.
- "The Top 300 of 2022".
- "Dextroamphetamine; Dextroamphetamine Saccharate; Amphetamine; Amphetamine Aspartate Drug Usage Statistics, United States, 2013 - 2022".
- "Commonly Abused Prescription Drugs Chart". National Institute on Drug Abuse.
- "Stimulant ADHD Medications – Methylphenidate and Amphetamines". National Institute on Drug Abuse.
- (2015). "Neuronal reward and decision signals: from theories to data". Physiological Reviews.
- "National Institute on Drug Abuse. 2009. Stimulant ADHD Medications – Methylphenidate and Amphetamines". National Institute on Drug Abuse.
- (2018). "Canadian ADHD Practice Guidelines". Canadian ADHD Resource Alliance.
- (May 2008). "Abuse of medications employed for the treatment of ADHD: results from a large-scale community survey". Medscape Journal of Medicine.
- (November 2013). "Contextual conditioning enhances the psychostimulant and incentive properties of d-amphetamine in humans". Addiction Biology.
- (27 February 2022). "Adderall- dextroamphetamine saccharate, amphetamine aspartate, dextroamphetamine sulfate, and amphetamine sulfate tablet".
- (3 March 2022). "Adderall XR- dextroamphetamine sulfate, dextroamphetamine saccharate, amphetamine sulfate and amphetamine aspartate capsule, extended release".
- (April 2008). "SPECT and PET of the dopamine transporter in attention-deficit/hyperactivity disorder". Expert Rev. Neurother..
- (February 2011). "How addictive drugs disrupt presynaptic dopamine neurotransmission". Neuron.
- (June 2002). "The role of zinc ions in reverse transport mediated by monoamine transporters". J. Biol. Chem..
- (October 2012). "Biomarkers and attention-deficit/hyperactivity disorder: a systematic review and meta-analyses". J. Am. Acad. Child Adolesc. Psychiatry.
- (April 2013). "A systematic review of combination therapy with stimulants and atomoxetine for attention-deficit/hyperactivity disorder, including patient characteristics, treatment strategies, effectiveness, and tolerability". J Child Adolesc Psychopharmacol.
- (2012). "Behavioral Neuroscience of Attention Deficit Hyperactivity Disorder and Its Treatment".
- (2009). "Atomoxetine attenuates dextroamphetamine effects in humans". Am J Drug Alcohol Abuse.
- (February 2015). "Behavioral, biological, and chemical perspectives on atypical agents targeting the dopamine transporter". Drug and Alcohol Dependence.
- (January 2001). "Amphetamine-type central nervous system stimulants release norepinephrine more potently than they release dopamine and serotonin". Synapse.
- (2013). "Powerful cocaine-like actions of 3,4-methylenedioxypyrovalerone (MDPV), a principal constituent of psychoactive 'bath salts' products". Neuropsychopharmacology.
- (22 May 2012). "Synthesis and Biological Evaluation of Rigid Analogues of Methamphetamines".
- (July 2008). "Dopamine Transporters: Chemistry, Biology and Pharmacology". Wiley.
- (2012). "The designer methcathinone analogs, mephedrone and methylone, are substrates for monoamine transporters in brain tissue". Neuropsychopharmacology.
- (October 2003). "Monoamine transporters and psychostimulant drugs". Eur J Pharmacol.
- (2006). "Therapeutic potential of monoamine transporter substrates". Current Topics in Medicinal Chemistry.
- (December 2001). "Amphetamine, 3,4-methylenedioxymethamphetamine, lysergic acid diethylamide, and metabolites of the catecholamine neurotransmitters are agonists of a rat trace amine receptor". Molecular Pharmacology.
- (January 2011). "The emerging role of trace amine-associated receptor 1 in the functional regulation of monoamine transporters and dopaminergic activity". Journal of Neurochemistry.
- (July 2001). "Trace amines: identification of a family of mammalian G protein-coupled receptors". Proceedings of the National Academy of Sciences of the United States of America.
- (January 2011). "VMAT2: a dynamic regulator of brain monoaminergic neuronal function interacting with drugs of abuse". Annals of the New York Academy of Sciences.
- (2010). "Goodman & Gilman's Pharmacological Basis of Therapeutics". McGraw-Hill.
- (December 2011). "Trace amine-associated receptor 1 is a stereoselective binding site for compounds in the amphetamine class". Bioorg. Med. Chem..
- "Dexedrine".
- "Dextroamphetamine [monograph]".
- (31 August 2013). "Information on Dexedrine: A Quick Review {{pipe}} Weitz & Luxenberg". Weitzlux.com.
- (June 2013). "Amphetamine, past and present—a pharmacological and clinical perspective". Journal of Psychopharmacology.
- (4 January 2017). "Prescription Forgery". Handwriting Services International.
- (January 1988). "Pharmaceutical Manufacturing Encyclopedia". Noyes Publications.
- "Dexedrine FAQs".
- (9 January 2003). "'Go pills': A war on drugs?". NBC News.
- "Air Force scientists battle aviator fatigue".
- (1995). "The use of amphetamines in U.S. Air Force tactical operations during Desert Shield and Storm". Aviation, Space, and Environmental Medicine.
- "Drugs@FDA: Dexedrine".
- "Drugs@FDA: Dexedrine".
- "Drugs@FDA: Dexedrine: Label and Approval History".
- (March 2011). "Bradley's Benzedrine studies on children with behavioral disorders". The Yale Journal of Biology and Medicine.
- "FDA Approved Drug Products: Label and Approval History (Benzedrine)".
- "National Drug Code Amphetamine Search Results". U.S. [[Food and Drug Administration]] (FDA).
- (28 October 2022). "Mydayis- dextroamphetamine sulfate, dextroamphetamine saccharate, amphetamine aspartate monohydrate, and amphetamine sulfate capsule, extended release".
- (10 March 2022). "Adzenys XR-ODT- amphetamine tablet, orally disintegrating".
- (27 January 2016). "Drug Approval Package: Adzenys XR-ODT (amphetamine)".
- (21 March 2022). "Drug Approval Package: Dyanavel XR".
- "Evekeo".
- "Zenzedi (dextroamphetamine sulfate, USP)". Zenzedi.com.
- "ProCentra (dextroamphetamine sulfate 5 mg/5 mL Oral Solution)". FSC Laboratories.
- "Abuse-resistant amphetamine prodrugs".
- (1995). "Stimulant treatment for attention deficit hyperactivity disorder". Australian Prescriber.
- "Pharmaceutical Services". .health.nsw.gov.au.
- "Dexamfetamine sulphate - Medicinal forms". BMJ Group and Pharmaceutical Press (Royal Pharmaceutical Society).
- (November 2010). "Dexamfetamine – Prescribe Generically". Interface Pharmacist Network Specialist Medicines (IPNSM).
- (December 2014). "Preclinical pharmacokinetics, pharmacology and toxicology of lisdexamfetamine: a novel d-amphetamine pro-drug". Neuropharmacology.
- (2006). "NRP-104 (lisdexamphetamine dimesylate)". U.S. Food and Drug Administration.
- (September 2011). "Advances and considerations in attention-deficit/hyperactivity disorder pharmacotherapy". Acta Medica Iranica.
- (October 2013). "A preclinical evaluation of the discriminative and reinforcing properties of lisdexamfetamine in comparison to D-amfetamine, methylphenidate and modafinil". Neuropharmacology.
- (November 2012). "Lisdexamfetamine and immediate release d-amfetamine - differences in pharmacokinetic/pharmacodynamic relationships revealed by striatal microdialysis in freely-moving rats with simultaneous determination of plasma drug concentrations and locomotor activity". Neuropharmacology.
- (July 2013). "A systematic review of psychostimulant treatment of negative symptoms of schizophrenia: challenges and therapeutic opportunities". Schizophrenia Research.
- (April 1988). "Dextro-amphetamine diminishes negative symptoms in schizophrenia". International Clinical Psychopharmacology.
- (July 2017). "Amphetamine Enhances Gains in Auditory Discrimination Training in Adult Schizophrenia Patients". Schizophrenia Bulletin.
This article was imported from Wikipedia and is available under the Creative Commons Attribution-ShareAlike 4.0 License. Content has been adapted to SurfDoc format. Original contributors can be found on the article history page.
Ask Mako anything about Dextroamphetamine — get instant answers, deeper analysis, and related topics.
Research with MakoFree with your Surf account
Create a free account to save articles, ask Mako questions, and organize your research.
Sign up freeThis content may have been generated or modified by AI. CloudSurf Software LLC is not responsible for the accuracy, completeness, or reliability of AI-generated content. Always verify important information from primary sources.
Report