From Surf Wiki (app.surf) — the open knowledge base
Cyproterone acetate
Chemical compound
_tablets_in_Australia,_with_three_blister_packs.jpg)
Chemical compound
| Drugs.com =
Free: 7% • Cyproterone (minor) • Acetic acid (minor) | elimination_half-life = Oral: 1.6–4.3 days IM: 3–4.3 days Urine: 30%
2098-66-0 (free alcohol)
Cyproterone acetate (CPA), sold alone under the brand name Androcur or with ethinylestradiol under the brand names Diane or Diane-35 among others, is an antiandrogen and progestin medication used in the treatment of androgen-dependent conditions such as acne, excessive body hair growth, early puberty, and prostate cancer, as a component of feminizing hormone therapy for transgender individuals, and in birth control pills. It is formulated and used both alone and in combination with an estrogen. CPA is taken by mouth one to three times per day.
_tablets_in_Australia,_with_three_blister_packs.jpg)
Common side effects of high-dose CPA in men include gynecomastia (breast development) and feminization. In both men and women, possible side effects of CPA include low sex hormone levels, reversible infertility, sexual dysfunction, fatigue, depression, weight gain, and elevated liver enzymes. With prolonged use, brain tumors prompting surgery are common, from 5% at high doses to 2% at low doses. At very high doses in older individuals, significant cardiovascular complications can occur. Rare but serious adverse reactions of CPA include blood clots, and liver damage. CPA can also cause adrenal insufficiency as a withdrawal effect if it is discontinued abruptly from a high dosage. CPA blocks the effects of androgens such as testosterone in the body, which it does by preventing them from interacting with their biological target, the androgen receptor (AR), and by reducing their production by the gonads, hence their concentrations in the body. In addition, it has progesterone-like effects by activating the progesterone receptor (PR). It can also produce weak cortisol-like effects at very high doses.
CPA was discovered in 1961. It was originally developed as a progestin. In 1965, the antiandrogenic effects of CPA were discovered. CPA was first marketed, as an antiandrogen, in 1973, and was the first antiandrogen to be introduced for medical use. A few years later, in 1978, CPA was introduced as a progestin in a birth control pill. It has been described as a "first-generation" progestin and as the prototypical antiandrogen. CPA is available widely throughout the world. An exception is the United States, where it is not approved for use.
Medical uses
CPA is used as a progestin and antiandrogen in hormonal birth control and in the treatment of androgen-dependent conditions. Specifically, CPA is used in combined birth control pills, in the treatment of androgen-dependent skin and hair conditions such as acne, seborrhea, excessive hair growth, and scalp hair loss, high androgen levels, in transgender hormone therapy, to treat prostate cancer, to reduce sex drive in sex offenders or men with paraphilias or hypersexuality, to treat early puberty, and for other uses. Treatment dosages range from 2mg or less, to 100mg or more daily.
In the United States, where CPA is not available, other medications with antiandrogenic effects are used to treat androgen-dependent conditions instead. Examples of such medications include gonadotropin-releasing hormone modulators (GnRH modulators) like leuprorelin and degarelix, nonsteroidal antiandrogens like flutamide and bicalutamide, the diuretic and steroidal antiandrogen spironolactone, the progestin medroxyprogesterone acetate, and the 5α-reductase inhibitors finasteride and dutasteride. The steroidal antiandrogen and progestin chlormadinone acetate is used as an alternative to CPA in Japan, South Korea, and a few other countries.
In 2020, the European Medicine Agency issued a warning that high doses of cyproterone acetate may contribute to risk of meningioma, and recommended physicians use alternative treatment for most indications (or the minimum effective dose where no alternatives were available) with the exception of prostate carcinoma.
Birth control
CPA is used with ethinylestradiol as a combined birth control pill to prevent pregnancy. This birth control combination has been available since 1978. The formulation is taken once daily for 21 days, followed by a 7-day free interval. CPA has also been available in combination with estradiol valerate (brand name Femilar) as a combined birth control pill in Finland since 1993. High-dose CPA tablets have a contraceptive effect and can be used as a form of birth control, although they are not specifically licensed as such.
Skin and hair conditions
Females
CPA is used as an antiandrogen to treat androgen-dependent skin and hair conditions such as acne, seborrhea, hirsutism (excessive hair growth), scalp hair loss, and hidradenitis suppurativa in women. These conditions are worsened by the presence of androgens, and by suppressing androgen levels and blocking their actions, CPA improves the symptoms of these conditions. CPA is used to treat such conditions both at low doses as a birth control pill and on its own at higher doses. A birth control pill containing low-dose CPA in combination with ethinylestradiol to treat acne has been found to result in overall improvement in 75 to 90% of women, with responses approaching 100% improvement. High-dose CPA alone likewise has been found to improve symptoms of acne by 75 to 90% in women. Discontinuation of CPA has been found to result in marked recurrence of symptoms in up to 70% of women. CPA is one of the most commonly used medications in the treatment of hirsutism, hyperandrogenism, and polycystic ovary syndrome in women throughout the world.
Higher dosages of CPA are used in combination with an estrogen specifically at doses of 25 to 100 mg/day cyclically in the treatment of hirsutism in women. The efficacy of such dosages of CPA in the treatment of hirsutism in women appear to be similar to that of spironolactone, flutamide, and finasteride. Randomized controlled trials have found that higher dosages of CPA (e.g., 20 mg/day or 100 mg/day) added cyclically to a birth control pill containing ethinylestradiol and 2 mg/day CPA were no more effective or only marginally more effective in the treatment of severe hirsutism in women than the birth control pill alone. Maintenance therapy with lower doses of CPA, such as 25 mg/day, has been found to be effective in preventing relapse of symptoms of hirsutism. CPA has typically been combined with ethinylestradiol, but it can alternatively be used in combination with hormone replacement therapy dosages of estradiol instead. CPA at a dosage of 50 mg/day in combination with 100 μg/day transdermal estradiol patches has been found to be effective in the treatment of hirsutism similarly to the combination of CPA with ethinylestradiol.
The efficacy of the combination of an estrogen and CPA in the treatment of hirsutism in women appears to be due to marked suppression of total and free androgen levels as well as additional blockade of the androgen receptor.
Males
CPA has been found to be effective in the treatment of acne in males, with marked improvement in symptoms observed at dosages of 25, 50, and 100 mg/day in different studies. It can also halt further progression of scalp hair loss in men. Increased head hair and decreased body hair has been observed with CPA in men with scalp hair loss. However, its side effects in men, such as demasculinization, gynecomastia, sexual dysfunction, bone density loss, and reversible infertility, make the use of CPA in males impractical in most cases. In addition, lower dosages of CPA, such as 25 mg/day, have been found to be better-tolerated in men. But such doses also show lower effectiveness in the treatment of acne in men.
High androgen levels
CPA is used as an antiandrogen to treat high androgen levels and associated symptoms such as masculinization due to conditions like polycystic ovary syndrome (PCOS) and congenital adrenal hyperplasia (CAH) in women. It is almost always combined with an estrogen, such as ethinylestradiol, when it is used in the treatment of PCOS in women.
Menopausal hormone therapy
CPA is used at low doses in menopausal hormone therapy in combination with an estrogen to provide endometrial protection and treat menopausal symptoms. It is used in menopausal hormone therapy under the brand name Climen, which is a sequential preparation that contains 2 mg estradiol valerate and 1 mg CPA. Climen was the first product for use in menopausal hormone therapy containing CPA to be marketed. It is available in more than 40 countries.
Transgender hormone therapy
CPA is widely used as an antiandrogen and progestogen in feminizing hormone therapy for transgender individuals. It has been historically used orally at a dosage of 10 to 100 mg/day and by intramuscular injection at a dosage of 300 mg once every 4 weeks. Many transgender individuals seeking feminizing hormone therapy have breast growth as one of the goals for undergoing feminizing hormone therapy, making this particular side effect of CPA generally viewed as a beneficial outcome rather than an issue.
Studies have found that 10, 25, 50, and 100 mg/day CPA in combination with estrogen all result in equivalent and full testosterone suppression in transgender women. In light of risks of CPA such as fatigue, blood clots, benign brain tumors, and liver damage, the use of lower dosages of CPA may help to minimize such risks. As a result, a CPA dosage of 10mg/day and no greater is now recommended by the World Professional Association for Transgender Health (WPATH) Standards of Care for the Health of Transgender and Gender Diverse People, Version 8 (SOC8).
CPA has an advantage over spironolactone as an antiandrogen in transgender people, as the combination of estrogen and CPA consistently suppresses testosterone levels into the normal female range whereas estrogen with spironolactone does not. Spironolactone is the most widely used antiandrogen in transgender women in the United States, whereas CPA is widely used in Europe and throughout the rest of the world.
Aside from adult transgender people, CPA has also been used as a puberty blocker and hence as an antiandrogen and antiestrogen to suppress puberty in transgender adolescents, although GnRH modulators are primarily used and more effective for this purpose.
Prostate cancer
CPA is used as an antiandrogen monotherapy and means of androgen deprivation therapy in the palliative treatment of prostate cancer in men. It is used at very high doses by mouth or by intramuscular injection to treat this disease. Antiandrogens do not cure prostate cancer, but can significantly extend life in men with the disease. CPA has similar effectiveness to GnRH modulators and surgical castration, high-dose estrogen therapy (e.g., with diethylstilbestrol), and high-dose nonsteroidal antiandrogen monotherapy (e.g., with bicalutamide), but has significantly inferior effectiveness to combined androgen blockade with a GnRH modulator and a nonsteroidal antiandrogen (e.g., with bicalutamide or enzalutamide). In addition, the combination of CPA with a GnRH modulator or surgical castration has not been found to improve outcomes relative to a GnRH modulator or surgical castration alone, in contrast to nonsteroidal antiandrogens. Due to its inferior effectiveness, tolerability, and safety, CPA is rarely used in the treatment of prostate cancer today, having largely been superseded by GnRH modulators and nonsteroidal antiandrogens. CPA is the only steroidal antiandrogen that continues to be used in the treatment of prostate cancer.
Dose-ranging studies of CPA for prostate cancer were not performed, and the optimal dosage of CPA for the treatment of the condition has not been established. A dosage range of oral CPA of 100 to 300 mg/day is used in the treatment of prostate cancer, but generally 150 to 200 mg/day oral CPA is used. Schröder (1993, 2002) reviewed the issue of CPA dosage and recommended a dosage of 200 to 300 mg/day for CPA as a monotherapy and a dosage of 100 to 200 mg/day for CPA in combined androgen blockade (that is, CPA in combination with surgical or medical castration). However, the combination of CPA with castration for prostate cancer has been found to significantly decrease overall survival compared to castration alone. Hence, the use of CPA as the antiandrogen component in combined androgen blockade would appear not to be advisable. When used by intramuscular injection to treat prostate cancer, CPA is used at a dosage of 300 mg once a week.
The combination of CPA with an estrogen such as ethinylestradiol sulfonate or low-dose diethylstilbestrol has been used as a form of combined androgen blockade and as an alternative to the combination of CPA with surgical or medical castration.
Sexual deviance
CPA is used as an antiandrogen and form of chemical castration in the treatment of paraphilias and hypersexuality in men. It is used to treat sex offenders. The medication is approved in more than 20 countries for this indication and is predominantly employed in Canada, Europe, and the Middle East. CPA works by decreasing sex drive and sexual arousal and producing sexual dysfunction. CPA can also be used to reduce sex drive in individuals with inappropriate sexual behaviors, such as people with intellectual disability and dementia. The medication is also used to reduce sexual behavior diagnosed as self-harmful, such as masochism. CPA has comparable effectiveness to medroxyprogesterone acetate in suppressing sexual urges and function but appears to be less effective than GnRH modulators like leuprorelin and has more side effects.
High-dose CPA significantly decreases sexual fantasies and sexual activity in 80 to 90% of men with paraphilias. In addition, it has been found to decrease the rate of reoffending in sex offenders from 85% to 6%, with most of the reoffenses being committed by individuals who did not follow their CPA treatment prescription. It has been reported that in 80% of cases, 100 mg/day CPA is adequate to achieve the desired reduction of sexuality, whereas in the remaining 20% of cases, 200 mg/day is sufficient. When only a partial reduction in sexuality is desired, 50 mg/day CPA can be useful. Reduced sexual desire and erectile function occurs with CPA by the end of the first week of treatment, and becomes maximal within three to four weeks. The dosage range is 50 to 300 mg/day.
Early puberty
CPA is used as an antiandrogen and antiestrogen to treat precocious puberty in boys and girls. However, it is not fully satisfactory for this indication because it is not able to completely suppress puberty. It does not suppress skeletal maturation enough to avoid a reduction in height at adulthood. For this reason, CPA has mostly been superseded by GnRH agonists in the treatment of precocious puberty. CPA is not satisfactory for gonadotropin-independent precocious puberty. CPA has been used at dosages of 50 to 300 mg/m2 to treat precocious puberty.
Other uses
CPA is useful in the treatment of hot flashes, for instance due to androgen deprivation therapy for prostate cancer.
CPA is useful for suppressing the testosterone flare at the initiation of GnRH agonist therapy. It has been used successfully both alone and in combination with estrogens such as diethylstilbestrol for this purpose.
Available forms
CPA is available in the form of oral tablets alone (higher-dose; 10 mg, 50 mg, 100 mg) or in combination with ethinylestradiol or estradiol valerate (low-dose; 1 or 2 mg CPA) and in the form of ampoules for intramuscular injection (higher-dose; 100 mg/mL, 300 mg/3 mL; brand name Androcur Depot).
The higher-dose formulations are used to treat prostate cancer and certain other androgen-related indications while the low-dose formulations which also have an estrogen are used as combined birth control pills and are used in menopausal hormone therapy for the treatment of menopausal symptoms.
Contraindications
Contraindications of CPA include:
- Hypersensitivity to CPA or any of the other components of the medication
- Pregnancy, lactation, and breastfeeding
- Puberty (except if being used to treat precocious puberty or delay puberty)
- Liver diseases and liver dysfunction
- Chronic kidney disease
- Dubin–Johnson syndrome and Rotor syndrome
- History of jaundice or persistent pruritus during pregnancy
- History of herpes during pregnancy
- Previous or existing liver tumors (only if not due to metastases from prostate cancer)
- Previous or existing meningioma, hyperprolactinemia, or prolactinoma
- Wasting syndromes (except in inoperable prostate cancer)
- Severe depression
- Previous or existing thromboembolic processes, as well as stroke and myocardial infarction
- Severe diabetes with vascular changes
- Sickle-cell anemia
When CPA is used in combination with an estrogen, contraindications for birth control pills should also be considered.
Side effects
Main article: Side effects of cyproterone acetate
CPA is generally well-tolerated and has a mild side-effect profile regardless of dosage when it is used in combination with an estrogen in women. Side effects of CPA in general include hypogonadism (low sex-hormone levels) and associated symptoms such as demasculinization, sexual dysfunction, infertility, and osteoporosis (fragile bones); breast changes such as breast tenderness, breast enlargement, and gynecomastia (breasts in men); emotional changes such as fatigue and depression; and other side effects such as vitamin B12 deficiency, weak glucocorticoid effects, and elevated liver enzymes. Weight gain can occur with CPA when it is used at high doses. Some of the side effects of CPA can be improved or fully prevented if it is combined with an estrogen to prevent estrogen deficiency. Few quantitative data are available on many of the potential side effects of CPA. Pooled tolerability data for CPA is not available in the literature. Cyproterone is also known to suppress adrenocortical function.
At very high doses in aged men with prostate cancer, CPA can cause cardiovascular side effects. Rarely, CPA can produce blood clots, liver toxicity (including hepatitis, liver failure, and liver cancer), excessively high prolactin levels, and certain benign brain tumors including meningiomas (tumors of the meninges) and prolactinomas (prolactin-secreting tumors of the pituitary gland). Upon discontinuation from high doses, CPA can produce adrenal insufficiency as a withdrawal effect.
Overdose
CPA is relatively safe in acute overdose. It is used at very high doses of up to 300 mg/day by mouth and 700 mg per week by intramuscular injection. For comparison, the dose of CPA used in birth control pills is 2 mg/day. There have been no deaths associated with CPA overdose. There are no specific antidotes for CPA overdose, and treatment should be symptom-based. Gastric lavage can be used in the event of oral overdose within the last 2 to 3 hours.
Interactions
Inhibitors and inducers of the cytochrome P450 enzyme CYP3A4 may interact with CPA. Examples of strong CYP3A4 inhibitors include ketoconazole, itraconazole, clotrimazole, and ritonavir, while examples of strong CYP3A4 inducers include rifampicin, rifampin, phenytoin, carbamazepine, phenobarbital, and St. John's wort. Certain anticonvulsant medications can substantially reduce levels of CPA, by as much as 8-fold.
Pharmacology
Pharmacodynamics
Main article: Pharmacology of cyproterone acetate#Pharmacodynamics
CPA has antiandrogenic activity, progestogenic activity, weak partial glucocorticoid activity, weak steroidogenesis inhibitor activity, and agonist activity at the pregnane X receptor. It has no estrogenic or antimineralocorticoid activity. In terms of potency, CPA is described as a highly potent progestogen, a moderately potent antiandrogen, and a weak glucocorticoid. Due to its progestogenic activity, CPA has antigonadotropic effects, and is able to suppress fertility and sex-hormone levels in both males and females.
Pharmacokinetics
Main article: Pharmacology of cyproterone acetate#Pharmacokinetics
CPA can be taken by mouth or by injection into muscle. It has near-complete oral bioavailability, is highly and exclusively bound to albumin in terms of plasma protein binding, is metabolized in the liver by hydroxylation and conjugation, has 15β-hydroxycyproterone acetate (15β-OH-CPA) as a single major active metabolite, has a long elimination half-life of about 2 to 4 days regardless of route of administration, and is excreted in feces primarily and to a lesser extent in urine.
Chemistry
CPA, also known as 1α,2α-methylene-6-chloro-17α-acetoxy-δ6-progesterone or as 1α,2α-methylene-6-chloro-17α-hydroxypregna-4,6-diene-3,20-dione acetate, is a synthetic pregnane steroid and an acetylated derivative of 17α-hydroxyprogesterone. It is structurally related to other 17α-hydroxyprogesterone derivatives such as chlormadinone acetate, hydroxyprogesterone caproate, medroxyprogesterone acetate, and megestrol acetate.
Synthesis
Chemical syntheses of CPA have been published. The following is one such synthesis:

The dehydrogenation of 17α-hydroxyprogesterone acetate [302-23-8] (1) with chloranil (tetrachloro-p-benzoquinone) gives a compound that has been called melengestrol acetate [425-51-4] (2). Dehydrogenation with selenium dioxide gives 17-acetoxy-1,4,6-pregnatriene-3,20-dione [2668-75-9] (3). Reacting this with diazomethane results in a 1,3-dipolar addition reaction at C1–C2 of the double bond of the steroid system, which forms a derivative of dihydropyrazole, CID:134990386 (4). This compound cleaves when reacted with perchloric acid, releasing nitrogen molecules and forming a cyclopropane derivative, 6-deschloro cyproterone acetate [2701-50-0] (5). Selective oxidation of the C6=C7 olefin with benzoyl peroxide gives the epoxide, i.e. 6-deschloro-6,7-epoxy cyproterone [15423-97-9] (6). The penultimate step involves a reaction with hydrochloric acid in acetic acid, resulting in the formation of chlorine and its subsequent dehydration, and a simultaneous opening of the cyclopropane ring giving 1α-(chloromethyl) chlormadinone acetate [17183-98-1] (7). The heating of this in collidine reforms the cyclopropane ring, completing the synthesis of CPA (8).
History
CPA was first synthesized in 1961 by Rudolf Wiechert, a Schering employee, and together with Friedmund Neumann in Berlin, they filed for a patent for CPA as "progestational agent" in 1962. The antiandrogenic activity of CPA was discovered serendipitously by Hamada, Neumann, and Karl Junkmann in 1963. Along with the steroidal antiandrogens benorterone (17α-methyl-B-nortestosterone; SKF-7690), cyproterone, BOMT (Ro 7–2340), and trimethyltrienolone (R-2956) and the nonsteroidal antiandrogens flutamide and DIMP (Ro 7–8117), CPA was one of the first antiandrogens to be discovered and researched.
CPA was initially developed as a progestogen for the prevention of threatened abortion. As part of its development, it was assessed for androgenic activity to ensure that it would not produce teratogenic effects in female fetuses. The drug was administered to pregnant rats and its effects on the rat fetuses were studied. To the surprise of the researchers, all of the rat pups born appeared to be female. After 20 female rat pups in a row had been counted, it was clear that this could not be a chance occurrence. The rat pups were further evaluated and it was found that, in terms of karyotype, about 50% were actually males. The male rat pups had been feminized, and this resultant finding constituted the discovery of the powerful antiandrogenic activity of CPA. A year after patent approval in 1965, Neumann published additional evidence of CPA's antiandrogenic effect in rats; he reported an "organizational effect of CPA on the brain". CPA started being used in animal experiments around the world to investigate how antiandrogens affected fetal sexual differentiation.
The first clinical use of CPA in the treatment of sexual deviance and prostate cancer occurred in 1966. It was first studied in the treatment of androgen-dependent skin and hair symptoms, specifically acne, hirsutism, seborrhea, and scalp hair loss, in 1969. CPA was first approved for medical use in 1973 in Europe under the brand name Androcur. In 1977, a formulation of CPA was introduced for use by intramuscular injection. CPA was first marketed as a birth control pill in 1978 in combination with ethinylestradiol under the brand name Diane. Following phase III clinical trials, CPA was approved for the treatment of prostate cancer in Germany in 1980. CPA became available in Canada as Androcur in 1987, as Androcur Depot in 1990, and as Diane-35 in 1998. Conversely, CPA was never introduced in any form in the United States. This was reportedly due to concerns about breast tumors observed with high-dose pregnane progestogens in beagle dogs as well as concerns about potential teratogenicity in pregnant women. Use of CPA in transgender women, an off-label indication, was reported as early as 1977. The use of CPA in transgender women was well-established by the early 1990s.
The history of CPA, including its discovery, development, and marketing, has been reviewed.
Society and culture
Generic names
The English and generic name of CPA is cyproterone acetate and this is its , , and . The English and generic name of unacetylated cyproterone is cyproterone and this is its and , while cyprotérone is the and French name and ciproterone is the and Italian name. The name of unesterified cyproterone in Latin is cyproteronum, in German is cyproteron, and in Spanish is ciproterona. These names of cyproterone correspond for CPA to acétate de cyprotérone in French, acetato de ciproterona in Spanish, ciproterone acetato in Italian, cyproteronacetat in German, siproteron asetat in Turkish, and cyproteronacetaat in Dutch. CPA is also known by the developmental code names SH-80714 and SH-714, while unacetylated cyproterone is known by the developmental code names SH-80881 and SH-881.
Brand names
CPA is marketed under brand names including Androcur, Androcur Depot, Androcur-100, Androstat, Asoteron, Cyprone, Cyproplex, Cyprostat, Cysaxal, Imvel, and Siterone. When CPA is formulated in combination with ethinylestradiol, it is also known as co-cyprindiol, and brand names for this formulation include Andro-Diane, Bella HEXAL 35, Chloe, Cypretil, Cypretyl, Cyproderm, Diane, Diane Mite, Diane-35, Dianette, Dixi 35, Drina, Elleacnelle, Estelle, Estelle-35, Ginette, Linface, Minerva, Vreya, and Zyrona. CPA is also marketed in combination with estradiol valerate as Climen, Climene, Elamax, and Femilar.
Availability
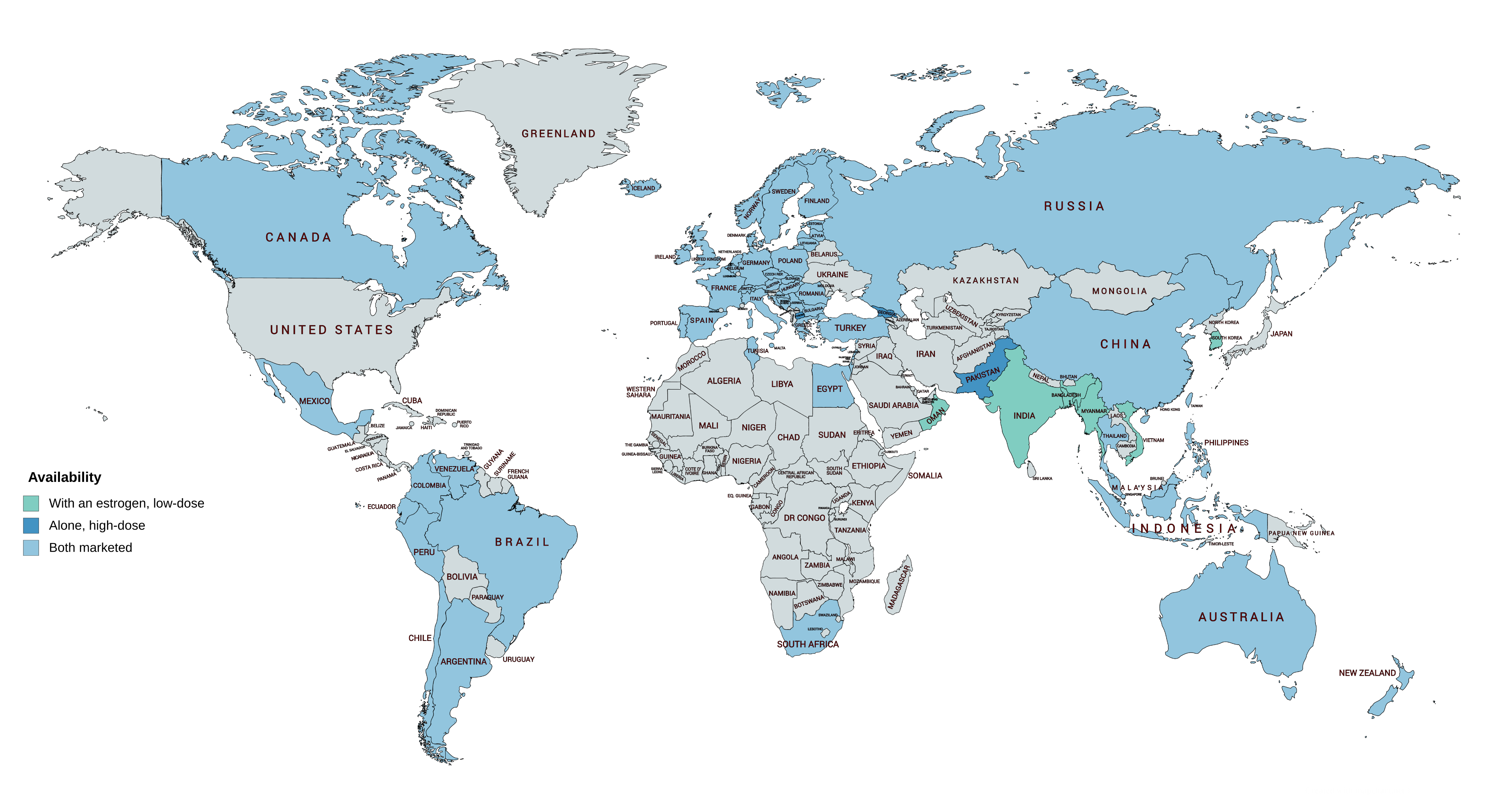
CPA is widely available throughout the world, and is marketed in almost every developed country, with the notable major exceptions of the United States and Japan. In almost all countries in which CPA is marketed, it is available both alone and in combination with an estrogen in birth control pills. CPA is marketed widely in combination with both ethinylestradiol and estradiol valerate. CPA-containing birth control pills are available in South Korea, but CPA as a standalone medication is not marketed in this country. In Japan and South Korea, the closely related antiandrogen and progestin chlormadinone acetate, as well as other medications, are used instead of CPA. Specific places in which CPA is marketed include the United Kingdom, elsewhere throughout Europe, Canada, Australia, New Zealand, South Africa, Latin America, and Asia. CPA is not marketed in most of Africa and the Middle East.
It has been said that the lack of availability of CPA in the United States explains why there are relatively few studies of it in the treatment of androgen-dependent conditions such as hyperandrogenism and hirsutism in women.
Generation
Progestins in birth control pills are sometimes grouped by generation. While the 19-nortestosterone progestins are consistently grouped into generations, the pregnane progestins that are or have been used in birth control pills are typically omitted from such classifications or are grouped simply as "miscellaneous" or "pregnanes". In any case, CPA has been described as a "first-generation" progestin similarly to closely related progestins like chlormadinone acetate, medroxyprogesterone acetate, and megestrol acetate.
Research
CPA has been studied and used in combination with low-dose diethylstilbestrol in the treatment of prostate cancer. The combination results in suppression of testosterone levels into the castrate range, which normally cannot be achieved with CPA alone. CPA has been studied as a form of androgen deprivation therapy for the treatment of benign prostatic hyperplasia (enlarged prostate). The medication has been studied in the treatment of breast cancer as well.
CPA has been studied for use as a potential male hormonal contraceptive both alone and in combination with testosterone in men. CPA was under development by Barr Pharmaceuticals in the 2000s for the treatment of hot flashes in prostate cancer patients in the United States. It reached phase III clinical trials for this indication and had the tentative brand name CyPat but development was ultimately discontinued in 2008. CPA is not satisfactorily effective as topical antiandrogen, for instance in the treatment of acne. CPA has been used to treat estrogen hypersensitivity vulvovaginitis in women.
CPA has been investigated for use in reducing aggression and self-injurious behavior via its antiandrogenic effects in conditions like autism spectrum disorders, dementias like Alzheimer's disease, and psychosis. CPA may be effective in the treatment of obsessive–compulsive disorder (OCD). CPA has been studied in the treatment of cluster headaches in men.
References
References
- (November 1988). "Pharmacokinetics of cyproterone acetate and its main metabolite 15 beta-hydroxy-cyproterone acetate in young healthy women". International Journal of Clinical Pharmacology, Therapy, and Toxicology.
- (June 2015). "Progestogens in menopausal hormone therapy". Przeglad Menopauzalny = Menopause Review.
- (December 2003). "Classification and pharmacology of progestins". Maturitas.
- (1 June 2002). "Systemic Drug Treatment in Dermatology: A Handbook". CRC Press.
- (October 1982). "Distribution and percentages of non-protein bound contraceptive steroids in human serum". Journal of Steroid Biochemistry.
- (27 September 2012). "Drugs in Palliative Care". OUP Oxford.
- (25 March 2010). "Pharmacology for Pharmacy and the Health Sciences: A Patient-centred Approach". OUP Oxford.
- (February 1985). "15-Hydroxycyproterone acetate and cyproterone acetate levels in plasma and urine". Journal of Chromatography.
- (22 July 2015). "Molecular Therapies of Cancer". Springer.
- (1998). "AAPL Newsletter". The Academy.
- (August 2005). "Pharmacology of estrogens and progestogens: influence of different routes of administration". Climacteric.
- (July 1994). "Cyproterone. A review of its pharmacology and therapeutic efficacy in prostate cancer". Drugs & Aging.
- (1994). "The antiandrogen cyproterone acetate: discovery, chemistry, basic pharmacology, clinical use and tool in basic research". Experimental and Clinical Endocrinology.
- (January 1977). "Pharmacology and potential use of cyproterone acetate". Hormone and Metabolic Research.
- (November 1986). "Pharmacology of antiandrogens". Journal of Steroid Biochemistry.
- (March 2024). "Use of progestogens and the risk of intracranial meningioma: national case-control study". BMJ.
- (2007). "Berek & Novak's Gynecology". Lippincott Williams & Wilkins.
- (December 1997). "Treatment of androgen excess in females: yesterday, today and tomorrow". Gynecological Endocrinology.
- (1 June 2002). "Systemic Drug Treatment in Dermatology: A Handbook". CRC Press.
- (25 February 2015). "Endocrinology: Adult and Pediatric E-Book". Elsevier Health Sciences.
- (2013). "Drug-Induced Liver Disease". Academic Press.
- (17 November 2011). "Hormonal Therapy for Male Sexual Dysfunction". John Wiley & Sons.
- EMA. European Medicines Agency. Assessment report cyproterone. 13 February 2020. https://www.ema.europa.eu/en/documents/referral/cyproterone-article-31-referral-prac-assessment-report_en.pdf. Accessed 9 Apr 2020.
- (28 January 2013). "Incapacitation: Trends and New Perspectives". Ashgate Publishing, Ltd..
- (6 December 2012). "ACNE and ROSACEA". Springer Science & Business Media.
- (September 2, 2015). "PrDIANE® -35".
- (May 2012). "An overview of the development of combined oral contraceptives containing estradiol: focus on estradiol valerate/dienogest". Gynecological Endocrinology.
- (January 2010). "Review of clinical experience with estradiol in combined oral contraceptives". Contraception.
- (November 2012). "Hormonal contraception in women at risk of vascular and metabolic disorders: guidelines of the French Society of Endocrinology". Annales d'Endocrinologie.
- (2003). "Cyproterone acetate for hirsutism". The Cochrane Database of Systematic Reviews.
- (June 2017). "The use of cyproterone acetate/ethinyl estradiol in hyperandrogenic skin symptoms - a review". The European Journal of Contraception & Reproductive Health Care.
- (1998). "Oral contraceptives and cyproterone acetate in female acne treatment". Dermatology.
- (May 1986). "Treatment of hirsutism and acne with cyproterone acetate". Clinics in Endocrinology and Metabolism.
- (October 2015). "Interventions for hidradenitis suppurativa". The Cochrane Database of Systematic Reviews.
- (2015). "Female pattern hair loss". Current Problems in Dermatology.
- (July 2012). "Combined oral contraceptive pills for treatment of acne". The Cochrane Database of Systematic Reviews.
- (September 1999). "Current aspects of antiandrogen therapy in women". Current Pharmaceutical Design.
- (July 2015). "Is hormonal treatment still an option in acne today?". The British Journal of Dermatology.
- (September 1988). "Anti-androgens in gynaecological practice". Bailliere's Clinical Obstetrics and Gynaecology.
- (January 1997). "Hirsutism". Lancet.
- (August 2000). "Idiopathic hirsutism". Endocrine Reviews.
- (May 2004). "Medical treatment regimens of hirsutism". Reproductive Biomedicine Online.
- (June 1999). "Unwanted body hair and its removal: a review". Dermatologic Surgery.
- (December 1986). "Clinical efficacy and safety of cyproterone acetate in severe hirsutism: results of a multicentered Canadian study". Fertility and Sterility.
- (July 1991). "Cyproterone acetate for severe hirsutism: results of a double-blind dose-ranging study". Clinical Endocrinology.
- (2005). "Hirsutism". Current Obstetrics & Gynaecology.
- (April 1991). "Treatment of hirsutism by an association of oral cyproterone acetate and transdermal 17 beta-estradiol". Fertility and Sterility.
- (July 1984). "Isotretinoin. A review of its pharmacological properties and therapeutic efficacy in acne and other skin disorders". Drugs.
- (1986). "Chapter 18. Chemical Control of Androgen Action". Academic Press.
- (1981). "Cyproteronacetate in the management of severe acne in males". Archives of Dermatological Research.
- (1986). "Response of male acne to antiandrogen therapy with cyproterone acetate". Dermatologica.
- (1989). "Pharmacology of the Skin II". Springer.
- (1 February 1995). "Hair Transplantation, Third Edition". Taylor & Francis.
- (July 1984). "Dermatologic therapy: December, 1982, through November, 1983". Journal of the American Academy of Dermatology.
- (2009). "Potential side effects of androgen deprivation treatment in sex offenders". The Journal of the American Academy of Psychiatry and the Law.
- (2012). "Aesthetic Medicine". Springer.
- (2002). "Managing cutaneous manifestations of hyperandrogenic disorders: the role of oral contraceptives". Treatments in Endocrinology.
- (June 2017). "Use of cyproterone acetate/ethinylestradiol in polycystic ovary syndrome: rationale and practical aspects". The European Journal of Contraception & Reproductive Health Care.
- (2012). "Hyperandrogenism in adolescent girls". Endocrine Development.
- (August 2009). "Pharmacological options for treatment of hyperandrogenic disorders". Mini Reviews in Medicinal Chemistry.
- (October 1998). "How actual is the treatment with antiandrogen alone in patients with polycystic ovary syndrome?". Journal of Endocrinological Investigation.
- (1997). "Women's Health and Menopause". Springer.
- (November 2003). "Androgens and antiandrogens". Annals of the New York Academy of Sciences.
- (December 2000). "The role of antiandrogens in hormone replacement therapy". Climacteric.
- (January 2008). "Long-term treatment of transsexuals with cross-sex hormones: extensive personal experience". The Journal of Clinical Endocrinology and Metabolism.
- (November 2017). "Endocrine Treatment of Gender-Dysphoric/Gender-Incongruent Persons: An Endocrine Society Clinical Practice Guideline". The Journal of Clinical Endocrinology and Metabolism.
- (April 2018). "Hormonal Treatment in Young People With Gender Dysphoria: A Systematic Review". Pediatrics.
- (17 June 2016). "Guidelines for the Primary and Gender-Affirming Care of Transgender and Gender Nonbinary People". Center of Excellence for Transgender Health.
- (29 September 2017). "Transsexual and Other Disorders of Gender Identity: A Practical Guide to Management". CRC Press.
- (20 May 2016). "Principles of Transgender Medicine and Surgery". Routledge.
- (March 2001). "Transgender Care: Recommended Guidelines, Practical Information, and Personal Accounts". Temple University Press.
- (2017). "Hormonbehandlung bei Transgenderpatienten". Gynäkologische Endokrinologie.
- (2009). "Behandlungsgrundsätze bei Transsexualität". Gynäkologische Endokrinologie.
- (September 2021). "Toward a Lowest Effective Dose of Cyproterone Acetate in Trans Women: Results From the ENIGI Study". The Journal of Clinical Endocrinology and Metabolism.
- (July 2021). "Low-Dose Cyproterone Acetate Treatment for Transgender Women". The Journal of Sexual Medicine.
- (February 2020). "Safety and rapid efficacy of guideline-based gender-affirming hormone therapy: an analysis of 388 individuals diagnosed with gender dysphoria". European Journal of Endocrinology.
- (2017). "Is a lower dose of cyproterone acetate as effective at testosterone suppression in transgender women as higher doses?". International Journal of Transgenderism.
- (19 August 2022). "Standards of Care for the Health of Transgender and Gender Diverse People, Version 8". International Journal of Transgender Health.
- (February 2023). "Gender-affirming hormonal therapy for transgender and gender-diverse people-A narrative review". Best Practice & Research. Clinical Obstetrics & Gynaecology.
- (June 2021). "MANAGEMENT OF ENDOCRINE DISEASE: Optimal feminizing hormone treatment in transgender people". European Journal of Endocrinology.
- (May 2021). "A systematic review of antiandrogens and feminization in transgender women". Clinical Endocrinology.
- (June 2023). "Feminizing gender-affirming hormone therapy for the transgender and gender diverse population: An overview of treatment modality, monitoring, and risks". Neurourology and Urodynamics.
- (July 2021). "Anti-Androgenic Effects Comparison Between Cyproterone Acetate and Spironolactone in Transgender Women: A Randomized Controlled Trial". The Journal of Sexual Medicine.
- (August 2020). "Differential Endocrine and Metabolic Effects of Testosterone Suppressive Agents in Transgender Women". Endocrine Practice.
- (July 2019). "Cyproterone acetate or spironolactone in lowering testosterone concentrations for transgender individuals receiving oestradiol therapy". Endocrine Connections.
- (September 2018). "Transgender medicine - puberty suppression". Reviews in Endocrine & Metabolic Disorders.
- (December 2014). "Approach to the patient: transgender youth: endocrine considerations". The Journal of Clinical Endocrinology and Metabolism.
- (October 2017). "Puberty suppression in transgender children and adolescents". The Lancet. Diabetes & Endocrinology.
- (May 2017). "Consecutive Cyproterone Acetate and Estradiol Treatment in Late-Pubertal Transgender Female Adolescents". The Journal of Sexual Medicine.
- (June 2005). "Cyproterone acetate in the therapy of prostate carcinoma". Archivio Italiano di Urologia, Andrologia.
- (1996). "Antiandrogens in Prostate Cancer".
- (December 1993). "Cyproterone acetate--mechanism of action and clinical effectiveness in prostate cancer treatment". Cancer.
- (1992). "The position of cyproterone acetate (CPA), a steroidal anti-androgen, in the treatment of prostate cancer". The Prostate. Supplement.
- (February 1991). "Use of cyproterone acetate in prostate cancer". The Urologic Clinics of North America.
- (2008). "Foye's Principles of Medicinal Chemistry". Lippincott Williams & Wilkins.
- (April 2000). "Single-therapy androgen suppression in men with advanced prostate cancer: a systematic review and meta-analysis". Annals of Internal Medicine.
- (29 September 2015). "Prostate Cancer: Science and Clinical Practice". Elsevier Science.
- (December 2004). "Androgen deprivation therapy for prostate cancer: current status and future prospects". The Prostate.
- (February 2007). "Antiandrogens in the treatment of prostate cancer". European Urology.
- (February 2000). "Androgen receptor antagonists (antiandrogens): structure-activity relationships". Current Medicinal Chemistry.
- (2008). "Management of advanced prostate cancer". Revista da Associação Médica Brasileira.
- (8 November 2010). "Cancer Chemotherapy and Biotherapy: Principles and Practice". Lippincott Williams & Wilkins.
- (11 February 2010). "Systemic Treatment of Prostate Cancer". OUP Oxford.
- (March 2003). "The role of antiandrogen monotherapy in the treatment of prostate cancer". BJU International.
- (2000). "Maximum androgen blockade in advanced prostate cancer: an overview of the randomised trials". The Lancet.
- (6 December 2012). "Antiandrogens in Prostate Cancer: A Key to Tailored Endocrine Treatment". Springer Science & Business Media.
- (2009). "Hormone Therapy in Breast and Prostate Cancer". Humana Press.
- (September 2007). "Combined androgen blockade in advanced prostate cancer: looking back to move forward". Clinical Genitourinary Cancer.
- (1996). "Antiandrogens in Prostate Cancer".
- (7 March 2013). "Prostatakarzinom — urologische und strahlentherapeutische Aspekte: urologische und strahlentherapeutische Aspekte". Springer-Verlag.
- (February 2015). "Pharmacological interventions for those who have sexually offended or are at risk of offending". The Cochrane Database of Systematic Reviews.
- (September 2015). "Androgen deprivation therapy (castration therapy) and pedophilia: What's new". Archivio Italiano di Urologia, Andrologia.
- (August 2000). "Protocols for the use of cyproterone, medroxyprogesterone, and leuprolide in the treatment of paraphilia". Canadian Journal of Psychiatry.
- (1991). "Cyproterone acetate in the treatment of sexual disorders: pharmacological base and clinical experience". Experimental and Clinical Endocrinology.
- (1980). "Therapeutic sex drive reduction". Acta Psychiatrica Scandinavica. Supplementum.
- (December 2008). "Pharmacology of sexually compulsive behavior". The Psychiatric Clinics of North America.
- (December 2008). "Inappropriate sexual behaviors in cognitively impaired older individuals". The American Journal of Geriatric Pharmacotherapy.
- (April 1989). "Antilibidinal drugs and mental retardation: a review". Medicine, Science, and the Law.
- (June 1985). "Self-harmful sexual behavior". The Psychiatric Clinics of North America.
- (June 2014). "Pharmacologic treatment of paraphilias". The Psychiatric Clinics of North America.
- (June 1975). "Antiandrogens in the treatment of sexual deviations of men". Journal of Steroid Biochemistry.
- (July 2000). "Experience with cyproterone acetate in the treatment of precocious puberty". Journal of Pediatric Endocrinology & Metabolism.
- (February 2008). "Update on the etiology, diagnosis and therapeutic management of sexual precocity". Arquivos Brasileiros de Endocrinologia e Metabologia.
- (31 August 2012). "Advances in Pediatrics". JP Medical Ltd.
- (March 1991). "Gonadotropin-independent precocious puberty". Endocrinology and Metabolism Clinics of North America.
- (January 2010). "Managing hot flushes in men after prostate cancer--a systematic review". Maturitas.
- (2003). "Incidence and management of hot flashes in prostate cancer". The Journal of Supportive Oncology.
- (1990). "Hot flushes: mechanism and prevention". Progress in Clinical and Biological Research.
- (2001). "Flare Associated with LHRH-Agonist Therapy". Reviews in Urology.
- (1 March 1999). "Textbook of Prostate Cancer: Pathology, Diagnosis and Treatment: Pathology, Diagnosis and Treatment". CRC Press.
- (2005). "Bicalutamide vs cyproterone acetate in preventing flare with LHRH analogue therapy for prostate cancer--a pilot study". Prostate Cancer and Prostatic Diseases.
- (October 1990). "Influence of different types of antiandrogens on luteinizing hormone-releasing hormone analogue-induced testosterone surge in patients with metastatic carcinoma of the prostate". The Journal of Urology.
- (1986). "Cyproterone acetate lead-in prevents initial rise of serum testosterone induced by luteinizing hormone-releasing hormone analogs in the treatment of metastatic carcinoma of the prostate". European Urology.
- (1988). "Endocrine Management of Prostatic Cancer". De Gruyter.
- (September 1993). "Luteinizing hormone-releasing hormone agonists in prostate cancer. Elimination of flare reaction by pretreatment with cyproterone acetate and low-dose diethylstilbestrol". Cancer.
- (2015). "Hirsutismus – Medikamentöse Therapie Gemeinsame Stellungnahme der Deutschen Gesellschaft für Gynäkologische Endokrinologie und Fortpflanzungsmedizin e.V. und des Berufsverbands der Frauenärzte e.V.". Journal für Reproduktionsmedizin und Endokrinologie.
- (19 June 1998). "European Drug Index: European Drug Registrations, Fourth Edition". CRC Press.
- (8 June 2012). "Dermatology E-Book". Elsevier Health Sciences.
- (2 July 2013). "Therapielexikon Dermatologie und Allergologie". Springer-Verlag.
- (1997). "Current Therapy in Adult Medicine". Mosby.
- "Androcur Label".
- "Mylan-Cyproterone Label".
- (1990). "Hair and Hair Diseases". Springer.
- (February 2010). "Anti-androgen treatments". Annales d'Endocrinologie.
- (December 1979). "Anti-androgen therapy in dermatology: a review". Clinical and Experimental Dermatology.
- (June 1999). "Antiandrogen treatment of polycystic ovary syndrome". Endocrinology and Metabolism Clinics of North America.
- (January 2009). "Drug treatment of paraphilic and nonparaphilic sexual disorders". Clinical Therapeutics.
- (December 1999). "Antiandrogens: a summary review of pharmacodynamic properties and tolerability in prostate cancer therapy". Archivio Italiano di Urologia, Andrologia.
- "Mylan-Cypersterone Monograph".
- (11 February 2009). "Sex Offenders: Identification, Risk Assessment, Treatment, and Legal Issues". Oxford University Press, USA.
- (2011). "Clinical Gynecologic Endocrinology and Infertility". Lippincott Williams & Wilkins.
- (14 September 2010). "Drug Management of Prostate Cancer". Springer.
- (May 2003). "Glucocorticoid receptor antagonism by cyproterone acetate and RU486". Molecular Pharmacology.
- (July 1987). "Inhibition of rat testicular 17 alpha-hydroxylase and 17,20-lyase activities by anti-androgens (flutamide, hydroxyflutamide, RU23908, cyproterone acetate) in vitro". Journal of Steroid Biochemistry.
- (6 January 2010). "Evaluation of Drug Candidates for Preclinical Development: Pharmacokinetics, Metabolism, Pharmaceutics, and Toxicology". John Wiley & Sons.
- (September 1998). "The human orphan nuclear receptor PXR is activated by compounds that regulate CYP3A4 gene expression and cause drug interactions". The Journal of Clinical Investigation.
- (December 2005). "Functional interactions between P-glycoprotein and CYP3A in drug metabolism". Expert Opinion on Drug Metabolism & Toxicology.
- (1974). "Androgens II and Antiandrogens / Androgene II und Antiandrogene". Springer.
- (6 December 2012). "Office Gynecology: Advanced Management Concepts". Springer Science & Business Media.
- (14 May 2014). "Pharmaceutical Substances: Syntheses, Patents and Applications of the most relevant APIs". Thieme.
- William Andrew Publishing. (22 October 2013). "Pharmaceutical Manufacturing Encyclopedia". Elsevier.
- "Further syntheses of cyproterone acetate".
- "Verfahren zur Herstellung von 1, 2alpha-Methylen-delta-17alpha-hydroxy-progesteronen".
- "6-chloro-1, 2alpha-methylene-delta6-17alpha-hydroxyprogesterone compounds and compositions".
- "Production of 1,2-methylene and 16,17-methylene Ketosteroids".
- "6-chloro-1, 2alpha-methylene-delta6-17alpha-hydroxyprogesterone compounds and compositions".
- (November 1963). "[Intrauterine Antimaskuline Beeinflussung von Rattenfeten Durch ein Stark Gestagen Wirksames Steroid]". Acta Endocrinologica.
- (6 December 2012). "Male Reproductive Function and Semen: Themes and Trends in Physiology, Biochemistry and Investigative Andrology". Springer Science & Business Media.
- (28 June 2014). "Hormones". Elsevier Science.
- (6 December 2012). "The Mechanism of Action of Androgens". Springer Science & Business Media.
- (December 1966). "Permanent changes in gonadal function and sexual behavior as a result of early feminization of male rats by treatment with an antiandrogenic steroid". Endokrinologie.
- (1971). "Use of androgen antagonists and antiandrogens in studies on sex differentiation". Gynecologic Investigation.
- (June 2010). "The World Federation of Societies of Biological Psychiatry (WFSBP) guidelines for the biological treatment of paraphilias". The World Journal of Biological Psychiatry.
- (October 1988). "Clinical applications of antiandrogens". Journal of Steroid Biochemistry.
- (April 1969). "[Management of hirsutism using cyproterone acetate]". Deutsche Medizinische Wochenschrift.
- (3 October 2014). "Cancer and its Management". Wiley.
- (1 December 1982). "Prostate Cancer". Williams & Wilkins.
- (June 1980). "Treatment of advanced prostatic cancer with parenteral cyproterone acetate: a phase III randomised trial". British Journal of Urology.
- (25 April 2012). "Androcur". Health Canada.
- (25 April 2012). "Androcur Depot". Health Canada.
- (25 April 2012). "DIANE-35". Health Canada.
- (2001). "Principles and Practice of Endocrinology and Metabolism". Lippincott Williams & Wilkins.
- (1977). "Of Homosexuality: The Current State of Knowledge". Journal of Christian Education.
- (1980). "Transsexualismus: Erfahrungen mit der operativen Korrektur bei männlichen Transsexuellen". Aktuelle Urologie.
- (1993). "Hormone Treatment in Transsexuals". Journal of Psychology & Human Sexuality.
- (March 1988). "[The antiandrogen cyproterone acetate. Its history from discovery to marketing]". Pharmazie in unserer Zeit.
- (14 November 2014). "The Dictionary of Drugs: Chemical Data: Chemical Data, Structures and Bibliographies". Springer.
- (6 December 2012). "Concise Dictionary of Pharmacological Agents: Properties and Synonyms". Springer Science & Business Media.
- (1 January 1988). "Dictionary of Obstetrics and Gynecology". Walter de Gruyter.
- "Cyproterone".
- (January 2000). "Index Nominum 2000: International Drug Directory". Taylor & Francis.
- (27 January 2015). "Acne: Causes and Practical Management". John Wiley & Sons.
- (22 September 2016). "Surgical Management of the Transgender Patient". Elsevier Health Sciences.
- http://www.micromedexsolutions.com/micromedex2/{{dead link. (July 2018)
- (2009). "Martindale: The Complete Drug Reference". Pharmaceutical Press.
- (11 July 2003). "Prostate Cancer: Science and Clinical Practice". Academic Press.
- (15 February 2000). "Contraceptive Choices and Realities: Proceedings of the 5th Congress of the European Society of Contraception". CRC Press.
- (2007). "Combined Estrogen-progestogen Contraceptives and Combined Estrogen-progestogen Menopausal Therapy". World Health Organization.
- (February 2017). "Progestins used in endocrine therapy and the implications for the biosynthesis and metabolism of endogenous steroid hormones". Molecular and Cellular Endocrinology.
- (2007). "Obstetrics, Gynecology & Infertility: Handbook for Clinicians". Scrub Hill Press, Inc..
- (December 1995). "Estrogens in the treatment of prostate cancer". The Journal of Urology.
- (December 1988). "The combination of cyproterone acetate and low dose diethylstilbestrol in the treatment of advanced prostatic carcinoma". The Journal of Urology.
- (June 1996). "Low-dose cyproterone acetate plus mini-dose diethylstilbestrol--a protocol for reversible medical castration". Urology.
- (January 1991). "Endocrine treatment of benign prostatic hypertrophy: current concepts". Urology.
- (1993). "[Androgen deprivation in benign prostatic hypertrophy]". Journal d'Urologie.
- (1993). "[Pharmacological combinations in the treatment of benign prostatic hypertrophy]". Journal d'Urologie.
- (June 1998). "Hormonal therapy of breast cancer". Cancer Treatment Reviews.
- (March 1988). "Clinical and endocrine effects of cyproterone acetate in postmenopausal patients with advanced breast cancer". European Journal of Cancer & Clinical Oncology.
- (November 2010). "Clinical trials in male hormonal contraception". Contraception.
- (2012). "Testosterone". Cambridge University Press.
- "Cyproterone acetate - Barr Laboratories - AdisInsight".
- (February 2018). "Cyproterone acetate in the treatment of oestrogen hypersensitivity vulvovaginitis". The Australasian Journal of Dermatology.
- (2012). "An evaluation of the role and treatment of elevated male hormones in autism spectrum disorders". Acta Neurobiologiae Experimentalis.
- (January 2011). "Cyproterone to treat aggressivity in dementia: a clinical case and systematic review". Journal of Psychopharmacology.
- (1993). "Aggression in humans: what is its biological foundation?". Neuroscience and Biobehavioral Reviews.
- (August 1991). "A possible antiaggressive effect of cyproterone acetate". The British Journal of Psychiatry.
- (December 2019). "Anti-Androgen Drugs in the Treatment of Obsessive-Compulsive Disorder: A Systematic Review". Current Medicinal Chemistry.
- (1 January 1989). "Obsessive-compulsive Disorder in Children and Adolescents". American Psychiatric Pub.
- (2010). "Drug treatment of obsessive-compulsive disorder". Dialogues in Clinical Neuroscience.
- (2004). "Images of Spanish Psychiatry". Editorial Glosa, S.L..
- (1988). "Antiandrogenic medication of cluster headache". International Journal of Clinical Pharmacology Research.
This article was imported from Wikipedia and is available under the Creative Commons Attribution-ShareAlike 4.0 License. Content has been adapted to SurfDoc format. Original contributors can be found on the article history page.
Ask Mako anything about Cyproterone acetate — get instant answers, deeper analysis, and related topics.
Research with MakoFree with your Surf account
Create a free account to save articles, ask Mako questions, and organize your research.
Sign up freeThis content may have been generated or modified by AI. CloudSurf Software LLC is not responsible for the accuracy, completeness, or reliability of AI-generated content. Always verify important information from primary sources.
Report