From Surf Wiki (app.surf) — the open knowledge base
Chan rearrangement
Chemical reaction
Chemical reaction
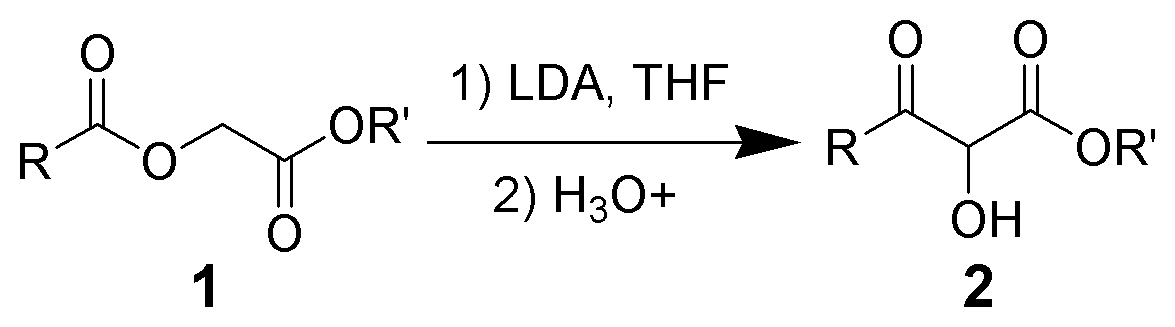
The Chan rearrangement is a chemical reaction that involves rearranging an acyloxy acetate (1) in the presence of a strong base to a 2-hydroxy-3-keto-ester (2).

This procedure was employed in the Holton Taxol total synthesis.
Reaction mechanism
The methylene bridge in the reactant with adjacent carbonyl and acetyl substituents is acidic and can be deprotonated by strong non-nucleophilic bases such as lithium tetramethylpiperidide or lithium diisopropylamide (LDA) as in an aldol reaction. The thus formed enolate then attacks the adjacent acetyl group through a short lived intermediate oxirane. Acidic workup liberates the free hydroxyl group.
References
- Rearrangement of α-acyloxyacetates into 2-hydroxy-3-ketoesters S. D. Lee, T. H. Chan, and K. S. Kwon Tetrahedron Lett. 1984, 25, 3399-3402. ()
- First total synthesis of taxol 1. Functionalization of the B ring Robert A. Holton, Carmen Somoza, Hyeong Baik Kim, Feng Liang, Ronald J. Biediger, P. Douglas Boatman, Mitsuru Shindo, Chase C. Smith, Soekchan Kim, et al.; J. Am. Chem. Soc. 1994, 116(4), 1597-1598. ()
This article was imported from Wikipedia and is available under the Creative Commons Attribution-ShareAlike 4.0 License. Content has been adapted to SurfDoc format. Original contributors can be found on the article history page.
Ask Mako anything about Chan rearrangement — get instant answers, deeper analysis, and related topics.
Research with MakoFree with your Surf account
Create a free account to save articles, ask Mako questions, and organize your research.
Sign up freeThis content may have been generated or modified by AI. CloudSurf Software LLC is not responsible for the accuracy, completeness, or reliability of AI-generated content. Always verify important information from primary sources.
Report