From Surf Wiki (app.surf) — the open knowledge base
Blanc chloromethylation
Chemical reaction
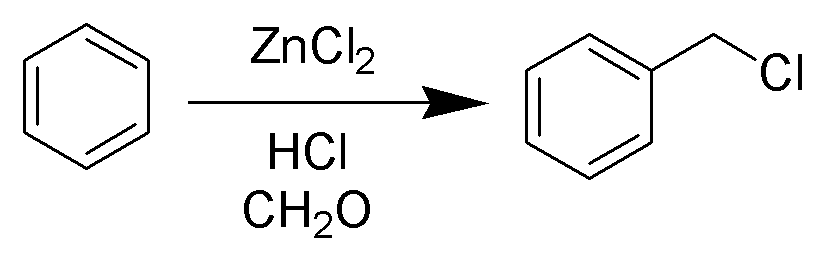
Chemical reaction
The Blanc chloromethylation (also called the Blanc reaction) is the chemical reaction of aromatic rings with formaldehyde and hydrogen chloride to form chloromethyl arenes. The reaction is catalyzed by Lewis acids such as zinc chloride. The reaction was discovered by Gustave Louis Blanc (1872-1927) in 1923.

Mechanism and scope
The reaction is carried out under acidic conditions and with a ZnCl2 catalyst. These conditions protonate the formaldehyde carbonyl making the carbon much more electrophilic. The aldehyde is then attacked by the aromatic pi-electrons, followed by rearomatization of the aromatic ring. The benzyl alcohol thus formed is quickly converted to the chloride under the reaction conditions.

Other possibilities for the electrophile include (chloromethyl)oxonium cation (ClH2C–OH2+) or chlorocarbenium cation (ClCH2+), which may be formed in the presence of zinc chloride. These species may account for the fact that moderately and strongly deactivated substrates that are inert to Friedel-Crafts reactions like acetophenone, nitrobenzene and p-chloronitrobenzene do show marginal reactivity of limited synthetic utility under chloromethylation conditions. Deactivated substrates give better results under modified chloromethylation conditions using chloromethyl methyl ether (MOMCl) in the presence of 60% H2SO4.
Highly activated arenes like phenols and anilines are not suitable substrates, since they undergo further electrophilic attack by Friedel-Crafts alkylation with the formed benzylic alcohol/chloride in an uncontrolled manner. In general, the formation of diarylmethane side product is a common outcome.
Although the reaction is an efficient means of introducing a chloromethyl group, the production of small amounts of highly carcinogenic bis(chloromethyl) ether is a disadvantage for industrial applications.
The corresponding fluoromethylation, bromomethylation and iodomethylation reactions can also be achieved, using the appropriate hydrohalic acid.
Additional reading
Safety
The reaction is performed with care as, like most chloromethylation reactions, it produces highly carcinogenic bis(chloromethyl) ether as a by-product.
References
References
- (30 September 1977). "New Data on the Chloromethylation of Aromatic and Heteroaromatic Compounds". [[Russian Chemical Reviews]].
- Blanc, Gustave Louis. (1923). "Sur la préparation de dérivés chlorométhyléniques aromatiques". [[Bulletin de la Société Chimique de France]].
- (1898). "Su alcuni derivati clorurati de trossimetilene". [[Gazzetta Chimica Italiana]].
- Laali, Kenneth K.. (2001). "Formaldehyde–Hydrogen Chloride". American Cancer Society.
- (1950-01-25). "Chloromethylation of Benzene Nucleus II". [[Yakugaku Zasshi]].
- (2011-03-15). "Organic Reactions". American Cancer Society.
- C., Norman, Richard O.. (2017). "Principles of Organic Synthesis". Routledge.
- D. Enders. (2002). "Diethyl [(Phenylsulfonyl)methyl]phosphonate". [[Organic Syntheses]].
- (2008). "Ion Exchangers".
This article was imported from Wikipedia and is available under the Creative Commons Attribution-ShareAlike 4.0 License. Content has been adapted to SurfDoc format. Original contributors can be found on the article history page.
Ask Mako anything about Blanc chloromethylation — get instant answers, deeper analysis, and related topics.
Research with MakoFree with your Surf account
Create a free account to save articles, ask Mako questions, and organize your research.
Sign up freeThis content may have been generated or modified by AI. CloudSurf Software LLC is not responsible for the accuracy, completeness, or reliability of AI-generated content. Always verify important information from primary sources.
Report