From Surf Wiki (app.surf) — the open knowledge base
Beta-peptide
Class of peptides derived from β-amino acids
Class of peptides derived from β-amino acids
Beta-peptides (β-peptides) are peptides derived from β-amino acids, in which the amino group is attached to the β-carbon (i.e. the carbon two atoms away from the carboxylate group). The parent β-amino acid is β-alanine (H2NCH2CH2CO2H), a common natural substance, but most examples feature substituents in place of one or more C-H bonds. β-peptides usually do not occur in nature. β-Peptide-based antibiotics are being explored as ways of evading antibiotic resistance. Early studies in this field were published in 1996 by the group of Dieter Seebach and that of Samuel Gellman.
Structure
As there are two carbon atoms available for substitution, β-amino acids have four sites (chirality included; as opposed to two in α-amino acids) for attaching the organic residue group.
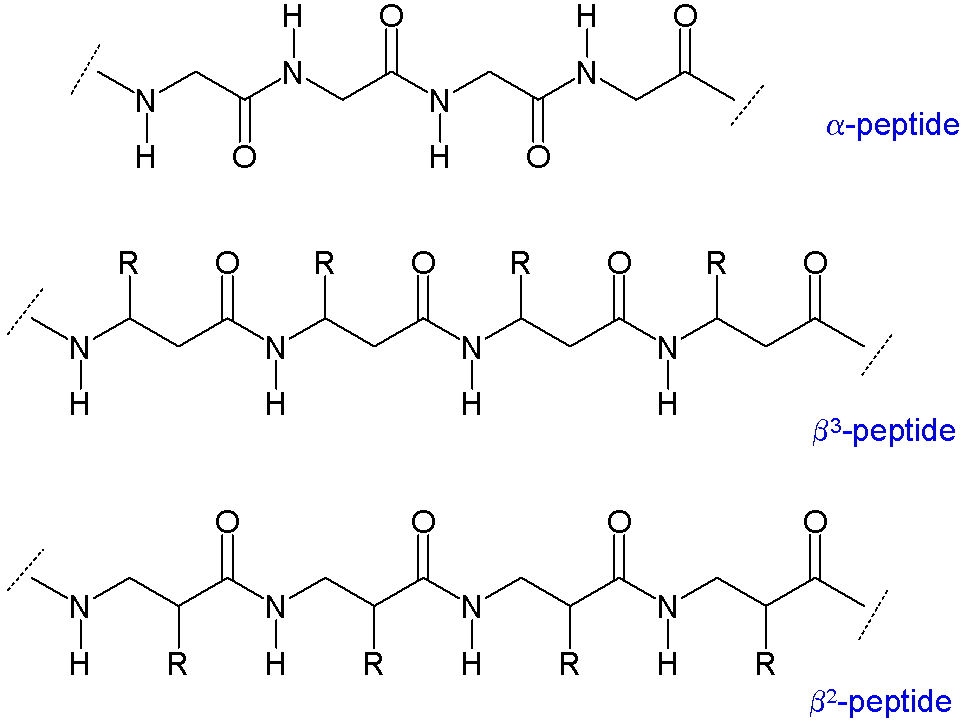
Synthesis
β-Amino acids have been prepared by many routes, including some based on the Arndt-Eistert synthesis.
Secondary structure
Because their backbones are longer than those of normal peptides, β-peptides form disparate secondary structures. The alkyl substituents at both the α and β positions in a β-amino acid favor a gauche conformation about the bond between the α-carbon and β-carbon. This also affects the thermodynamic stability of the structure.
Many types of helix structures consisting of β-peptides have been reported. These conformation types are distinguished by the number of atoms in the hydrogen-bonded ring that is formed in solution; 8-helix, 10-helix, 12-helix, 14-helix, and 10/12-helix have been reported. Generally speaking, β-peptides form a more stable helix than α-peptides.
Clinical potential
β-Peptides are stable against proteolytic degradation in vitro and in vivo, a potential advantage over natural peptides. β-Peptides have been used to mimic natural peptide-based antibiotics such as magainins, which are highly potent but difficult to use as drugs because they are degraded by proteolytic enzymes.
Examples
β-Amino acids with a wide variety of substituents exist. Named by analogy to the biological α-amino acids, the following have been found naturally: β-alanine, β-leucine, β-lysine, β-arginine, β-glutamate, β-glutamine, β-phenylalanine and β-tyrosine. Of these, β-alanine is found in mammals and incorporated in pantothenic acid, an essential nutrient. Aspartic acid is structurally a β-amino acids. Microcystins are a class of compounds containing a β-isoaspartyl (i.e. aspartic acid linked with its beta-carboxyl) residue.
References
References
- (June 1996). "β-Peptides: Synthesis by Arndt-Eistert homologation with concomitant peptide coupling. Structure determination by NMR and CD spectroscopy and by X-ray crystallography. Helical secondary structure of a -hexapeptide in solution and its stability towards pepsin". [[Helvetica Chimica Acta]].
- (1996). "β-Peptide Foldamers: Robust Helix Formation in a New Family of -Amino Acid Oligomers". [[J. Am. Chem. Soc.]].
- (1997). "β-Peptides: a surprise at every turn". [[Chem. Commun.]].
- (November 2005). "Conformationally constrained β-amino acid derivatives by intramolecular [2 + 2]-photocycloaddition of a tetronic acid amide and subsequent lactone ring opening". J. Org. Chem..
- (September 2005). "Efficient synthesis of a β-peptide combinatorial library with microwave irradiation". J. Am. Chem. Soc..
- (January 2017). "Homooligomeric β3 (R)-valine peptides: Transformation between C14 and C12 helical structures induced by a guest Aib residue". Biopolymers.
- (October 1999). "Beta-peptides: twisting and turning". Curr. Med. Chem..
- (January 2006). "Toward a rational design of β-peptide structures". J Comput Chem.
- (2002). "Mimicry of host-defense peptides by unnatural oligomers: antimicrobial β-peptides". [[J. Am. Chem. Soc.]].
- (6 May 2005). "Enantioselective Synthesis of Beta-Amino Acids". Wiley Inc..
This article was imported from Wikipedia and is available under the Creative Commons Attribution-ShareAlike 4.0 License. Content has been adapted to SurfDoc format. Original contributors can be found on the article history page.
Ask Mako anything about Beta-peptide — get instant answers, deeper analysis, and related topics.
Research with MakoFree with your Surf account
Create a free account to save articles, ask Mako questions, and organize your research.
Sign up freeThis content may have been generated or modified by AI. CloudSurf Software LLC is not responsible for the accuracy, completeness, or reliability of AI-generated content. Always verify important information from primary sources.
Report