From Surf Wiki (app.surf) — the open knowledge base
Adrenal tumor
Tumors of the adrenal gland

Tumors of the adrenal gland
| Field | Value |
|---|---|
| name | Adrenal tumor |
| image | Incidences and prognoses of adrenal tumors.png |
| caption | Incidences and prognoses of adrenal tumors. |
An adrenal tumor or adrenal mass is any benign or malignant neoplasms of the adrenal gland, several of which are notable for their ability to overproduce endocrine hormones. Adrenal cancer is the presence of malignant adrenal tumors, which include neuroblastoma, adrenocortical carcinoma and some adrenal pheochromocytomas. Most adrenal pheochromocytomas and all adrenocortical adenomas are benign tumors, which do not metastasize or invade nearby tissues, but may cause significant symptoms by dysregulating hormones.
Adrenal glands
Main article: Adrenal gland

Adrenal glands, also known as the suprarenal glands, are located above each kidney and in the retroperitoneal space. They produce and regulate different hormones, including stress hormones like cortisol and hormones that regulate salt and water balance like aldosterone. The glands are triangular-shaped and are composed of two types of tissues: the cortex and the medulla. The glands are surrounded by a capsule made up of connective tissue and consist of outer portion or the cortex, and the inner portion or the medulla. The outer cortex is further divided into three layers, named the zona glomerulosa, zona fasciculata and zona reticularis, all which produce critical steroid hormones.
The inner portion or the medulla produces catecholamines such as adrenaline (epinephrine) and norepinephrine. Mineralocorticoid hormones like aldosterone, which regulates blood pressure, kidney function, and certain sex hormones, are produced by the outer layer of the adrenal cortex or the zona glomerulosa.Glucocorticoids, which are produced by the middle layer of the adrenal cortex, or the zona fasciculata. Glucocorticoids are critical for regulation of blood sugar and the immune system, as well as response to physiological stress. Androgens, which aid in the development of secondary sex characteristics, are produced by the inner zona reticularis. DHEA (dehydroepiandrosterone) is the most commonly found hormone in the body and is a precursor in the production of progesterone, estrogen, cortisol and testosterone.
Tumors of the adrenal cortex
Adrenocortical adenoma
Main article: Adrenocortical adenoma
Adrenocortical adenomas are benign tumors of the adrenal cortex that are extremely common (present in 1–10% of persons at autopsy). They can be confused with adrenocortical "nodules", however nodules are not true neoplasms. Adrenocortical adenomas are uncommon in patients younger than 30 years old, and have equal incidence in both sexes. The clinical significance of these neoplasms is two-fold. First, they have been detected as incidental findings with increasing frequency in recent years, due to the increasing use of CT scans and magnetic resonance imaging in a variety of medical settings. This can result in additional testing and invasive procedures to rule out the slight possibility of an early adrenocortical carcinoma. Second, a minority (about 15%) of adrenocortical adenomas are "functional", meaning that they produce glucocorticoids, mineralocorticoids, and/or sex steroids, resulting in endocrine disorders such as Cushing's syndrome, Conn's syndrome (hyperaldosteronism), virilization of females, or feminization of males. Functional adrenocortical adenomas are surgically curable.
Most of the adrenocortical adenomas are less than 2 cm in greatest dimension and less than 50 grams in weight. However, size and weight of the adrenal cortical tumors are no longer considered to be a reliable sign of benignity or malignancy. Grossly, adrenocortical adenomas are encapsulated, well-circumscribed, solitary tumors with solid, homogeneous yellow-cut surface. Necrosis and hemorrhage are rare findings.
Adrenocortical carcinoma
Main article: Adrenocortical carcinoma
Adrenocortical carcinoma (ACC) is a rare, highly aggressive cancer of adrenal cortical cells, which may occur in children or adults. ACCs may be "functional", producing steroid hormones and consequent endocrine dysfunction similar to that seen in many adrenocortical adenomas. Due to their location deep in the retroperitoneum, most adrenocortical carcinomas are not diagnosed until they have grown quite large. Adrenocortical carcinomas frequently invade large vessels, such as the renal vein and inferior vena cava, as well as metastasize via the lymphatics and through the blood to the lungs and other organs.
The most effective treatment is surgery, although this is not feasible for many patients, and the overall prognosis of the disease is poor. Chemotherapy, radiation therapy, and hormonal therapy may also be employed in the treatment of this disease.
Tumors of the adrenal medulla
The adrenal medulla is located anatomically at the center of each adrenal gland, and is composed of neuroendocrine (chromaffin) cells which produce and release epinephrine (adrenaline) into the bloodstream in response to activation of the sympathetic nervous system. Neuroblastoma and pheochromocytoma are the two most important tumors which arise from the adrenal medulla. Both tumors may also arise from extra-adrenal sites, specifically, in the paraganglia of the sympathetic chain.
Neuroblastoma
Main article: Neuroblastoma
Neuroblastoma is an aggressive cancer of immature neuroblastic cells (precursors of neurons), and is one of the most common pediatric cancers, with a median age at diagnosis of two years. Adrenal neuroblastoma typically presents with a rapidly enlarging abdominal mass. Although the tumor has often spread to distant parts of the body at the time of diagnosis, this cancer is unusual in that many cases are highly curable when the spread is limited to the liver, skin, and/or bone marrow (stage IVS). Related, but less aggressive tumors composed of more mature neural cells include ganglioneuroblastoma and ganglioneuroma. Neuroblastic tumors often produce elevated levels of catecholamine hormone metabolites, such as vanillylmandelic acid (VMA) and homovanillic acid, and may produce severe watery diarrhea through production of vasoactive intestinal peptide. Treatment of neuroblastoma includes surgery and radiation therapy for localized disease, and chemotherapy for metastatic disease.
Pheochromocytoma
Main article: Pheochromocytoma
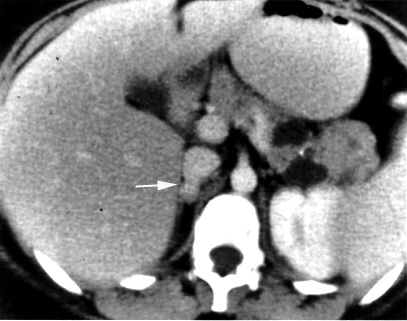
Pheochromocytoma is a neoplasm composed of cells similar to the chromaffin cells of the mature adrenal medulla. Pheochromocytomas occur in patients of all ages, and may be sporadic, or associated with a hereditary cancer syndrome, such as multiple endocrine neoplasia (MEN) types IIA and IIB, neurofibromatosis type I, or von Hippel–Lindau syndrome. Only 10% of adrenal pheochromocytomas are malignant, while the rest are benign tumors. The most clinically important feature of pheochromocytomas is their tendency to produce large amounts of the catecholamine hormones epinephrine (adrenaline) and norepinephrine. This may lead to potentially life-threatening high blood pressure, or cardiac arrhythmias, and numerous symptoms such as headache, palpitations, anxiety attacks, sweating, weight loss, and tremor. Diagnosis is most easily confirmed through urinary measurement of catecholamine metabolites such as VMA and metanephrines. Most pheochromocytomas are initially treated with anti-adrenergic drugs to protect against catecholamine overload, with surgery employed to remove the tumor once the patient is medically stable.
Incidentalomas
An adrenal incidentaloma is an adrenal tumor found by coincidence without clinical symptoms or suspicion. It is one of the more common unexpected findings revealed by computed tomography (CT), magnetic resonance imaging (MRI), or ultrasonography. Adrenal incidentalomas often secrete cortisol and require thorough clinical evaluation.
Management of incidentalomas include evaluation by a endocrinologist or endocrine surgeon. Tumors under 3 cm are generally considered benign and are only treated if there are grounds for a diagnosis of Cushing's syndrome or pheochromocytoma. Radiodensity helps with estimating the risk of malignancy in a tumor. A tumor of 10 Hounsfield units or less on an un-enhanced CT is likely a lipid-rich adenoma.
On CT scan, benign adenomas typically are of low radiographic density (due to fat content) and show rapid washout of contrast medium (50% or more of the contrast medium washes out at 10 minutes). If the hormonal evaluation is negative and imaging suggests benign, follow-up should be considered with imaging at 6, 12, and 24 months and repeat hormonal evaluation yearly for 4 years.
Adrenal-dedicated CT and MRI imaging can be performed to distinguish benign adenomas from potentially malignant lesions. Online calculators assist radiologists in calculating the washout of contrast in adrenal nodules on CT and chemical shift on MRI.
Hormonal evaluations can also be used, which include:
- 1-mg overnight dexamethasone suppression test to detect excess cortisol
- 24-hour urinary specimen for measurement of fractionated metanephrines and catecholamines
- Blood plasma aldosterone concentration and plasma renin activity, if hypertension is present
Metastasis to the adrenals
Metastasis, or mets, to one or both adrenal glands is the most common form of malignant adrenal lesion, and the second most common adrenal tumor after benign adenomas. Primary tumors in are most commonly from lung cancer (39%), breast cancer (35%), melanoma, gastrointestinal tract cancer, pancreas cancer, and renal cancer. Clinical concern for mets to adrenal glands requires evaluation for adrenal insufficiency as well as evaluation for tumor markers that can determine involvement of other areas of metastatic disease.
Epidemiology
Adrenal tumors are common tumors in humans. The detection of adrenal incidentalomas has increased, likely due to improvement in imaging modalities. A population based study by Ebbehoj et. al. determined a 10x increase in adrenal tumors from 1995 to 2010, aligning with an increase in abdominal scans. Incidentalomas were also determined to be increasingly functional due to better evaluation methods for hormonal imbalances that the tumors may cause. In review of literature by Zeiger et al., it was also determined that about 80% of adrenal tumors found incidentally were non-functional adenomas, while the remaining had functional, malignant or metastatic tumors.
Treatment
Surgical
Diagnosis of adrenal tumors occur through lab and imaging studies. For adrenal carcinomas, the most effective treatment is surgery, however, surgery may not be feasible for many patients and the overall prognosis of the disease is poor. Chemotherapy, radiation therapy, and hormonal therapy may also be employed in the treatment of this disease.
Because pheochromocytomas can cause imbalances in catecholamines that can be life-threatening, treatment and optimization before surgery is required. Close monitoring during the surgery and careful consideration for the anesthesia used during the surgery for a pheochromocytoma is also required to ensure that catecholamine release is avoided. Patients can also experience a drop in blood pressure or hypotension after the tumor is removed, which is treated with adrenergic agonists and fluids.
In pregnant patients, functional adrenal tumors can lead to complications in pregnancy, for example, if the tumor leads to symptoms of hypertension. Surgical management (adrenalectomy) has been determined to be safe for pregnant patients and fetuses.
A 2018 Cochrane Systematic review compared two different types of surgery: laparoscopic retroperitoneal adrenalectomy and laparoscopic transperitoneal adrenalectomy in different types of adrenal tumors. Laparoscopic retroperitoneal adrenalectomy appeared to reduce late morbidity, time to oral fluid or food intake and time to ambulation, when compared to laparoscopic transperitoneal adrenalectomy. However there was uncertainty in these findings due to low-quality evidence, as well as inconclusive findings about effects of either surgery on all-cause mortality, early morbidity, socioeconomic effects, duration of surgery, operative blood loss, and conversion to open surgery. Lymphadenectomy or removal of surrounding lymph nodes is also recommended.
Non-surgical management
Non-surgical management of adrenocortical cancer: Although surgery is recommended for the best chance of survival without reoccurrence, Mitotane has also been used in the non-surgical management of adrenocortical carcinomas as adjuvant therapy. Mitotane is given orally via tablets and levels are monitored to ensure proper levels. Retrospective studies have analyzed the efficacy of mitotane and have determined that it should be used as adjuvant therapy in cases of patients with increased risk of reoccurrence. Mitotane can induce hormonal dysregulation including adrenal insufficiency, hypothyroidism, and hypogonadism. Non-hormonal side effects of this medication include fatigue, GI symptoms, dyslipidemia, skin rash and mild leukopenia.
Future diagnostic tools
Blood circulating microRNAs (miRNA) has been investigated in the recent years for the potential as a less-invasive biomarker for adrenal diseases. Nine studies have so far investigated the occurrence of circulating miRNAs in blood from patients diagnosed with adrenocortical tumors. MiRNAs can be released into the blood stream by three different types of excretion. Cellular damage with passive release (necrosis, inflammation), active secretion in the form of extracellular vesicles (EV) (microvesicles, exosomes and apoptotic bodies) or in association with high density lipoproteins (HDL) and Argonaute (AGO) proteins. The majority of miRNAs found in blood are in complexes with AGO. Despite the promising results, the sensitivity of potential circulating miRNA markers for adrenocortical tumors appears to be variable. An increase in sensitivity could possibly be achieved by targeting only EV-associated miRNAs as the release of miRNAs into EVs are hypothesized to be a controlled process. EV-associated miRNAs could thereby act as more specific markers of malignancy. However, as there were significant differences in the results of the studies performed, methodological differences and low patient numbers could contribute to this discrepancy. This calls for further studies on larger cohorts with uniform methodological requirements to clarify the applicability of circulating miRNAs as biomarkers of prognosis and malignancy in patients diagnosed with adrenocortical tumors.
References
References
- "Doctors remove 'world's largest adrenal tumour'". The Hindu.
- (November 2020). "Nebennierentumore". Therapeutische Umschau.
- (2003-03-04). "Management of the Clinically Inapparent Adrenal Mass (Incidentaloma)". Annals of Internal Medicine.
- (2025). "Anatomy, Abdomen and Pelvis: Adrenal Glands (Suprarenal Glands)". StatPearls Publishing.
- Data and references for pie chart are located at [[Commons:File:Incidences and prognoses of adrenal tumors.png. file description page in Wikimedia Commons.]]
- (2021-07-28). "Neuroendocrine and Adrenal Tumors, Version 2.2021, NCCN Clinical Practice Guidelines in Oncology". Journal of the National Comprehensive Cancer Network.
- (2022). "Overview of the 2022 WHO Classification of Adrenal Cortical Tumors". Endocrine Pathology.
- (2025). "Adrenal Cancer". StatPearls Publishing.
- (2025). "Adrenal Adenoma". StatPearls Publishing.
- (2000). "Adrenal Incidentaloma". MDText.com, Inc..
- (1982-08-13). "Incidental Asymptomatic Adrenal Masses Detected by Computed Tomographic Scanning: Is Operation Required?". JAMA.
- (2014). "Adrenocortical carcinoma". Endocrine Reviews.
- (2018-10-01). "European Society of Endocrinology Clinical Practice Guidelines on the management of adrenocortical carcinoma in adults, in collaboration with the European Network for the Study of Adrenal Tumors". European Journal of Endocrinology.
- "Neuroblastoma".
- (2015). "Adrenal Glands: From Pathophysiology to Clinical Evidence". Nova Science.
- (20–26 August 2005). "Phaeochromocytoma". Lancet.
- (January 2003). "Evaluation and management of the incidental adrenal mass". Proceedings.
- (2020). "Epidemiology of adrenal tumours in Olmsted County, Minnesota, USA: a population-based cohort study". The Lancet. Diabetes & Endocrinology.
- (2009-07-01). "American Association Of Clinical Endocrinologists And American Association Of Endocrine Surgeons Medical Guidelines For The Management Of Adrenal Incidentalomas". Endocrine Practice.
- (2009-07-01). "American Association Of Clinical Endocrinologists And American Association Of Endocrine Surgeons Medical Guidelines For The Management Of Adrenal Incidentalomas". Endocrine Practice.
- (February 2019). "Distinguishing adrenal adenomas from non-adenomas with multidetector CT: evaluation of percentage washout values at a short time delay triphasic enhanced CT". The British Journal of Radiology.
- "Adrenal Incidentalomas: Biochemical Workup and Surgical Indications".
- (2020). "Technical and Interpretive Pitfalls in Adrenal Imaging". Radiographics.
- (2016). "Chemical Shift MR Imaging of the Adrenal Gland: Principles, Pitfalls, and Applications". Radiographics.
- "Adrenal Washout Calculator for CT".
- "Adrenal Chemical Shift MRI Calculator".
- (February 2007). "Clinical practice. The incidentally discovered adrenal mass". The New England Journal of Medicine.
- (2003-03-04). "Management of the Clinically Inapparent Adrenal Mass (Incidentaloma)". Annals of Internal Medicine.
- List of included entries and references is found on main image page in Commons: [[:File:Metastasis sites for common cancers.svg]]
- (2025). "Adrenal Metastasis". StatPearls Publishing.
- (2020-04-01). "Adrenal Incidentalomas are Tied to Increased Risk of Diabetes: Findings from a Prospective Study". The Journal of Clinical Endocrinology & Metabolism.
- (2025). "Perioperative Management of Pheochromocytoma". StatPearls Publishing.
- (2023). "Surgical treatment of adrenal tumors during pregnancy". Reviews in Endocrine & Metabolic Disorders.
- (December 2018). "Transperitoneal versus retroperitoneal laparoscopic adrenalectomy for adrenal tumours in adults". The Cochrane Database of Systematic Reviews.
- (2024-09-01). "A review of mitotane in the management of adrenocortical cancer". The Oncologist.
- (February 2020). "Non-Coding RNAs in Adrenocortical Cancer: From Pathogenesis to Diagnosis". Cancers.
- (2021-02-22). "Circulating microRNA Expression in Cushing's Syndrome". Frontiers in Endocrinology.
- (April 2015). "Suggested roles for microRNA in tumors". Biomolecular Concepts.
- (November 2012). "Cell-to-cell miRNA transfer: from body homeostasis to therapy". Pharmacology & Therapeutics.
- (March 2012). "Circulating microRNAs: a novel class of biomarkers to diagnose and monitor human cancers". Medicinal Research Reviews.
- (March 2011). "Argonaute2 complexes carry a population of circulating microRNAs independent of vesicles in human plasma". Proceedings of the National Academy of Sciences of the United States of America.
- (September 2011). "Characterization of extracellular circulating microRNA". Nucleic Acids Research.
- (June 2007). "Exosome-mediated transfer of mRNAs and microRNAs is a novel mechanism of genetic exchange between cells". Nature Cell Biology.
This article was imported from Wikipedia and is available under the Creative Commons Attribution-ShareAlike 4.0 License. Content has been adapted to SurfDoc format. Original contributors can be found on the article history page.
Ask Mako anything about Adrenal tumor — get instant answers, deeper analysis, and related topics.
Research with MakoFree with your Surf account
Create a free account to save articles, ask Mako questions, and organize your research.
Sign up freeThis content may have been generated or modified by AI. CloudSurf Software LLC is not responsible for the accuracy, completeness, or reliability of AI-generated content. Always verify important information from primary sources.
Report