From Surf Wiki (app.surf) — the open knowledge base
Acetoacetic ester synthesis
Chemical reaction
Chemical reaction
Acetoacetic ester synthesis is a chemical reaction where ethyl acetoacetate is alkylated at the α-carbon to both carbonyl groups and then converted into a ketone, or more specifically an α-substituted acetone. This is very similar to malonic ester synthesis.
Mechanism
A strong base deprotonates the dicarbonyl α-carbon. This carbon is preferred over the methyl carbon because the formed enolate is conjugated and thus resonance stabilized. The carbon then undergoes nucleophilic substitution. When heated with aqueous acid, the newly alkylated ester is hydrolyzed to a β-keto acid, which is decarboxylated to form a methyl ketone. The alkylated ester can undergo a second substitution to produce the dialkylated product.
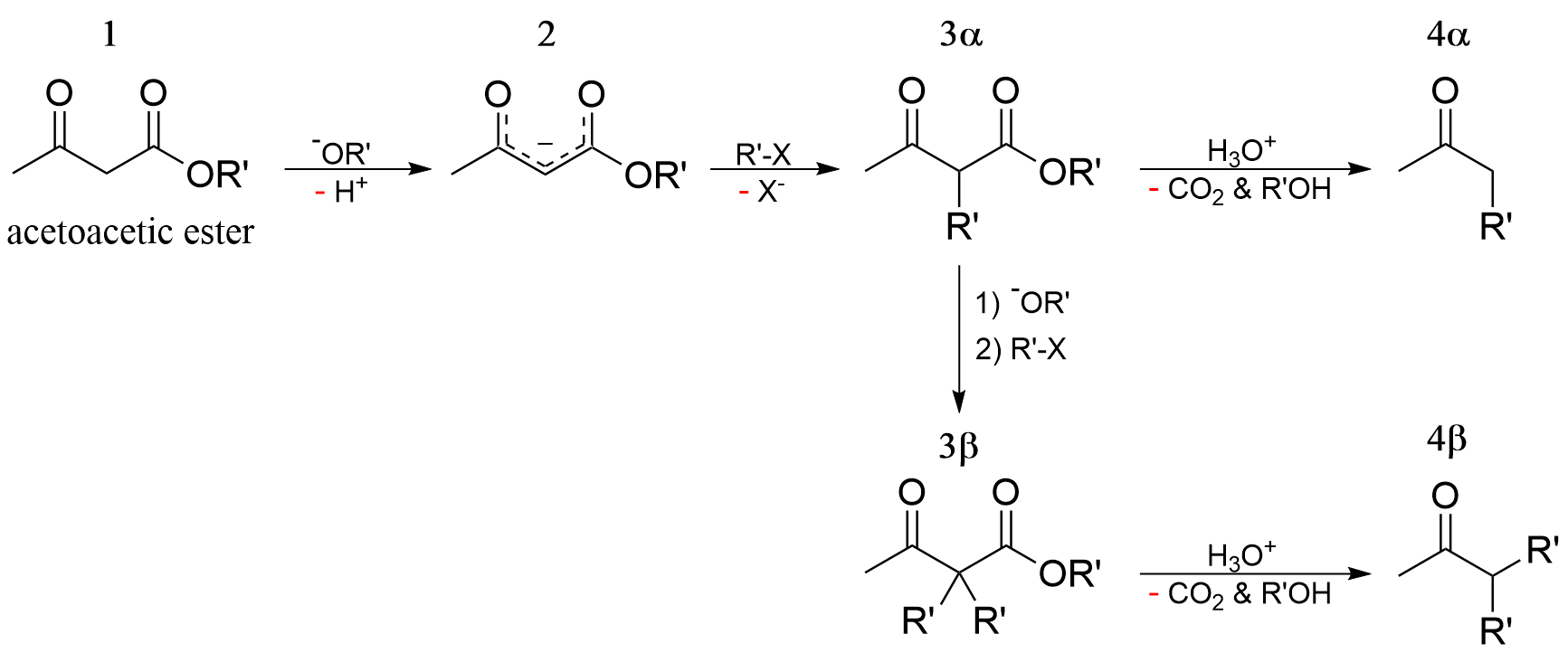
Double deprotonation of ethyl acetoacetate
The classical acetoacetatic ester synthesis utilizes the 1:1 conjugate base. Ethyl acetoacetate is however diprotic: :CH3C(O)CH2CO2Et + NaH → CH3C(O)CH(Na)CO2Et + H2 :CH3C(O)CH(Na)CO2Et + BuLi → LiCH2C(O)CH(Na)CO2Et + BuH
The dianion (i.e., LiCH2C(O)CH(Na)CO2Et) adds electrophile to the terminal carbon as depicted in the following simplified form: :LiCH2C(O)CH(Na)CO2Et + RX → RCH2C(O)CH(Na)CO2Et + LiX
References
References
- Smith, Janice Gorzynski. ''Organic Chemistry: Second Ed''. 2008. pp 905–906
- [https://pharmaxchange.info/press/2011/02/acetoacetic-ester-synthesis-alkylation-of-enolates/ Acetoacetic Ester Synthesis – Alkylation of Enolates. PharmaXChange.info]
- (2007). "Stereoselective Isoprenoid Chain Extension with Acetoacetate Dianion: [(''E, E, E'')-Geranylgeraniol from (''E, E'')-Farnesol". Organic Syntheses.
This article was imported from Wikipedia and is available under the Creative Commons Attribution-ShareAlike 4.0 License. Content has been adapted to SurfDoc format. Original contributors can be found on the article history page.
Ask Mako anything about Acetoacetic ester synthesis — get instant answers, deeper analysis, and related topics.
Research with MakoFree with your Surf account
Create a free account to save articles, ask Mako questions, and organize your research.
Sign up freeThis content may have been generated or modified by AI. CloudSurf Software LLC is not responsible for the accuracy, completeness, or reliability of AI-generated content. Always verify important information from primary sources.
Report