From Surf Wiki (app.surf) — the open knowledge base
Dakin–West reaction
Reaction that converts amino acids into a keto-amides
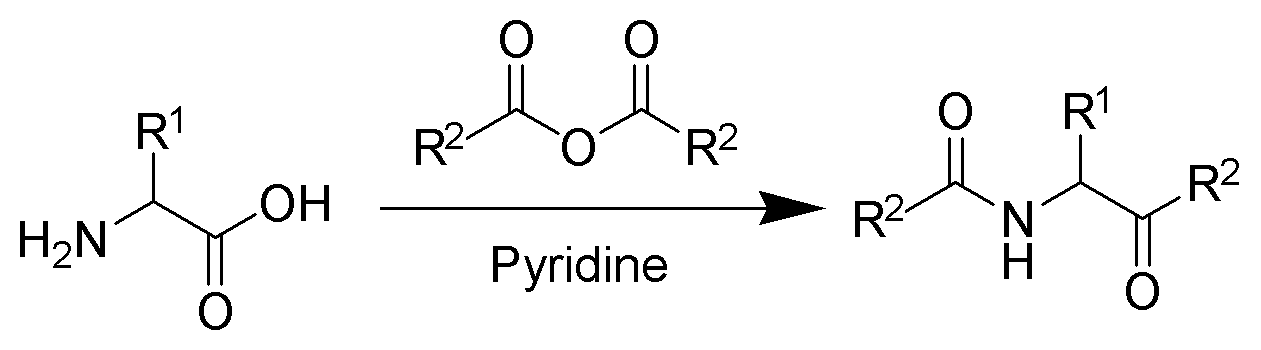
Reaction that converts amino acids into a keto-amides
The Dakin–West reaction is a chemical reaction that transforms an amino-acid into a keto-amide using an acid anhydride and a base, typically pyridine. It is named for Henry Drysdale Dakin and Randolph West. In 2016 Schreiner and coworkers reported the first asymmetric variant of this reaction employing short oligopeptides as catalysts.

With pyridine as a base and solvent, refluxing conditions are required. However, with the addition of 4-dimethylaminopyridine (DMAP) as a catalyst, the reaction can take place at room temperature.
With some acids, this reaction can take place even in the absence of an α-amino group.
This reaction should not be confused with the Dakin reaction.
Reaction mechanism
The reaction mechanism involves the acylation and activation of the acid 1 to the mixed anhydride 3. The amide will serve as a nucleophile for the cyclization forming the azlactone 4. Deprotonation and acylation of the azlactone forms the key carbon-carbon bond. Subsequent ring-opening of 6 and decarboxylation give the final keto-amide product.

General ketone synthesis
Modern variations on the Dakin–West reaction permit many enolizable carboxylic acids – not merely amino acids – to be converted to their corresponding methyl ketones. For example, β-aryl carboxylic acids can be efficiently converted to β-aryl ketones by treatment of an acetic anhydride solution of the acid with catalytic N-methylimidazole. This reactivity is attributed in part to generation of acetylimidazolium, a powerful cationic acetylating agent, in situ.

References
References
- (1928). "A General Reaction of Amino Acids". The Journal of Biological Chemistry.
- (1928). "A General Reaction of Amino Acids. II". The Journal of Biological Chemistry.
- (1928). "Some Aromatic Derivatives of Substituted Acetylaminoacetones". The Journal of Biological Chemistry.
- (1947). "The Conversion of Amino Acids to Oxazoles". The Journal of Organic Chemistry.
- (1988). "The Dakin–West reaction". Chemical Society Reviews.
- (2016). "The Enantioselective Dakin-West Reaction". Angewandte Chemie International Edition.
- (1953). "3-Acetamido-2-butanone". Organic Syntheses.
- (1978). "4-Dialkylaminopyridines as Highly Active Acylation Catalysts. [New synthetic method (25)]". Angewandte Chemie International Edition in English.
- (1970). "Zum Mechanismus der Dakin-West-Reaktion, I Die Reaktion von N-Acyl-sek.-aminosäuren mit Acetanhydrid". Chemische Berichte.
- (1974). "Kinetic and mechanistic studies of the Dakin-West reaction". The Journal of Organic Chemistry.
- (2006). "Dakin−West Synthesis of β-Aryl Ketones". The Journal of Organic Chemistry.
This article was imported from Wikipedia and is available under the Creative Commons Attribution-ShareAlike 4.0 License. Content has been adapted to SurfDoc format. Original contributors can be found on the article history page.
Ask Mako anything about Dakin–West reaction — get instant answers, deeper analysis, and related topics.
Research with MakoFree with your Surf account
Create a free account to save articles, ask Mako questions, and organize your research.
Sign up freeThis content may have been generated or modified by AI. CloudSurf Software LLC is not responsible for the accuracy, completeness, or reliability of AI-generated content. Always verify important information from primary sources.
Report